Crystallization of sodium acetate
Bildnummer 13502073
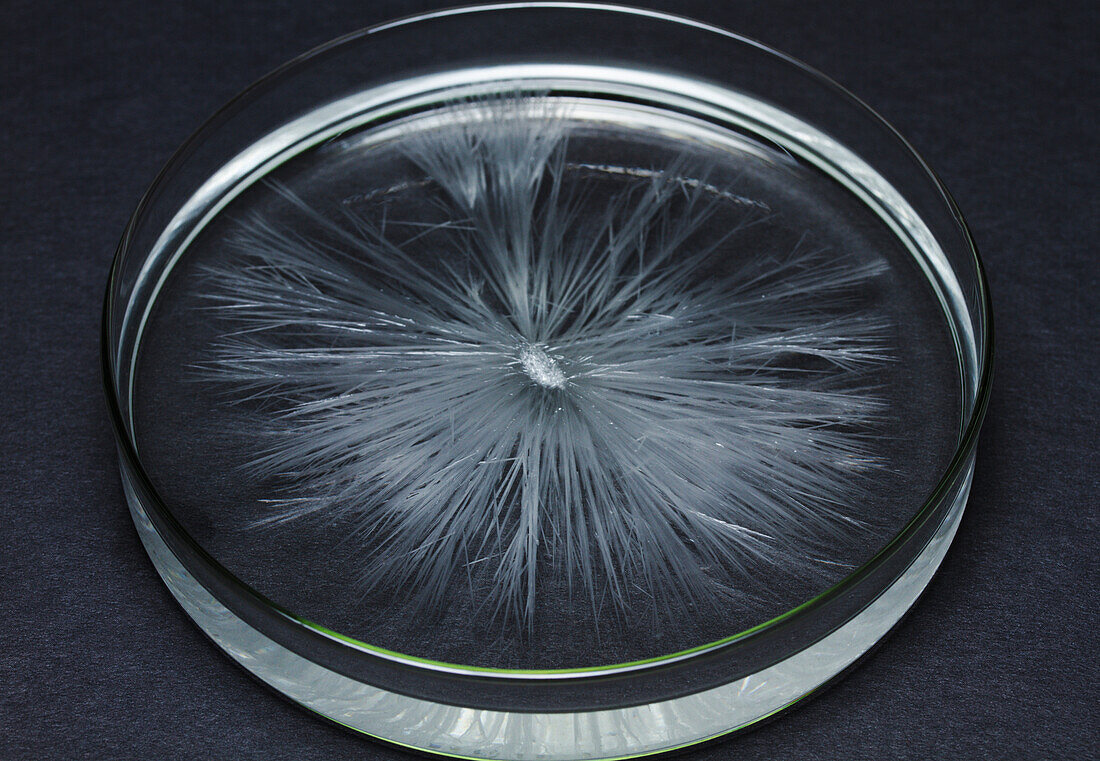
| Crystallization of sodium acetate. When solid sodium acetate trihydrate (CH3COONa 3H2O) melts at 58C, it releases bound water (water of crystallization) and dissolves in it. When the solution is then allowed to cool slowly, it becomes supersaturated. When a nucleation centre (a small piece of solid sodium acetate) is introduced, it causes the solution to crystallize back into solid sodium acetate trihydrate. This process is exothermic and releases heat. It found its use in consumer heating pads. | |
| Lizenzart: | Lizenzpflichtig |
| Credit: | Science Photo Library / SCIENCE SOURCE / Turtle Rock Scientific |
| Bildgröße: | 5070 px × 3509 px |
| Modell-Rechte: | nicht erforderlich |
| Eigentums-Rechte: | nicht erforderlich |
| Restrictions: | - |
Preise für dieses Bild ab 15 €
Universitäten & Organisationen
(Informationsmaterial Digital, Informationsmaterial Print, Lehrmaterial Digital etc.)
ab 15 €
Redaktionell
(Bücher, Bücher: Sach- und Fachliteratur, Digitale Medien (redaktionell) etc.)
ab 30 €
Werbung
(Anzeigen, Aussenwerbung, Digitale Medien, Fernsehwerbung, Karten, Werbemittel, Zeitschriften etc.)
ab 55 €
Handelsprodukte
(bedruckte Textilie, Kalender, Postkarte, Grußkarte, Verpackung etc.)
ab 75 €
Pauschalpreise
Rechtepakete für die unbeschränkte Bildnutzung in Print oder Online
ab 495 €
