Model of ethene before hybridisation RHS
Bildnummer 13417306
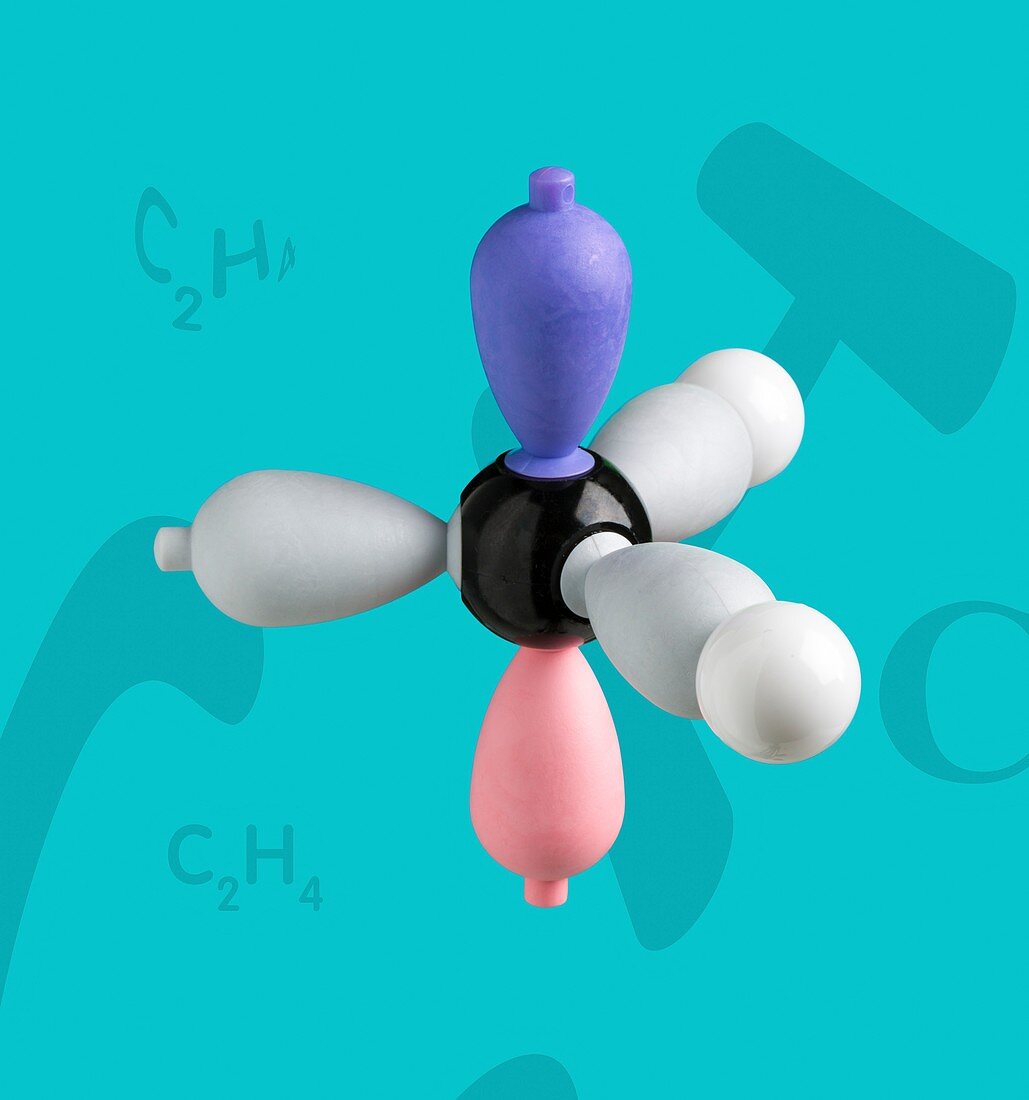
| Electron density model of ethene before hybridisation Right hand side of Ethene model before hybridization. Ethene is a very important hydrocarbon which has the formula C2H4. The model shows sigma- and pi-bonding orbitals, and the concept of hybridisation and delocalisation. A carbon and two hydrogen atoms form the left and right sides of the simplest alkene compound - ethene. In an sp2 hybridization, one s orbital is mixed with two p orbitals to form three sp2 hybridized orbitals. Much of the production of ethene goes to polyethylene manufacture and also as a plant hormone to speed up the ripening of fruit. | |
| Lizenzart: | Lizenzpflichtig |
| Credit: | Science Photo Library / Chillmaid, Martyn F. |
| Bildgröße: | 4044 px × 4321 px |
| Modell-Rechte: | nicht erforderlich |
| Eigentums-Rechte: | nicht erforderlich |
| Restrictions: | - |
Preise für dieses Bild ab 15 €
Universitäten & Organisationen
(Informationsmaterial Digital, Informationsmaterial Print, Lehrmaterial Digital etc.)
ab 15 €
Redaktionell
(Bücher, Bücher: Sach- und Fachliteratur, Digitale Medien (redaktionell) etc.)
ab 30 €
Werbung
(Anzeigen, Aussenwerbung, Digitale Medien, Fernsehwerbung, Karten, Werbemittel, Zeitschriften etc.)
ab 55 €
Handelsprodukte
(bedruckte Textilie, Kalender, Postkarte, Grußkarte, Verpackung etc.)
ab 75 €
Pauschalpreise
Rechtepakete für die unbeschränkte Bildnutzung in Print oder Online
ab 495 €
