Rutherford scattering experiment, illustration
Bildnummer 13376930
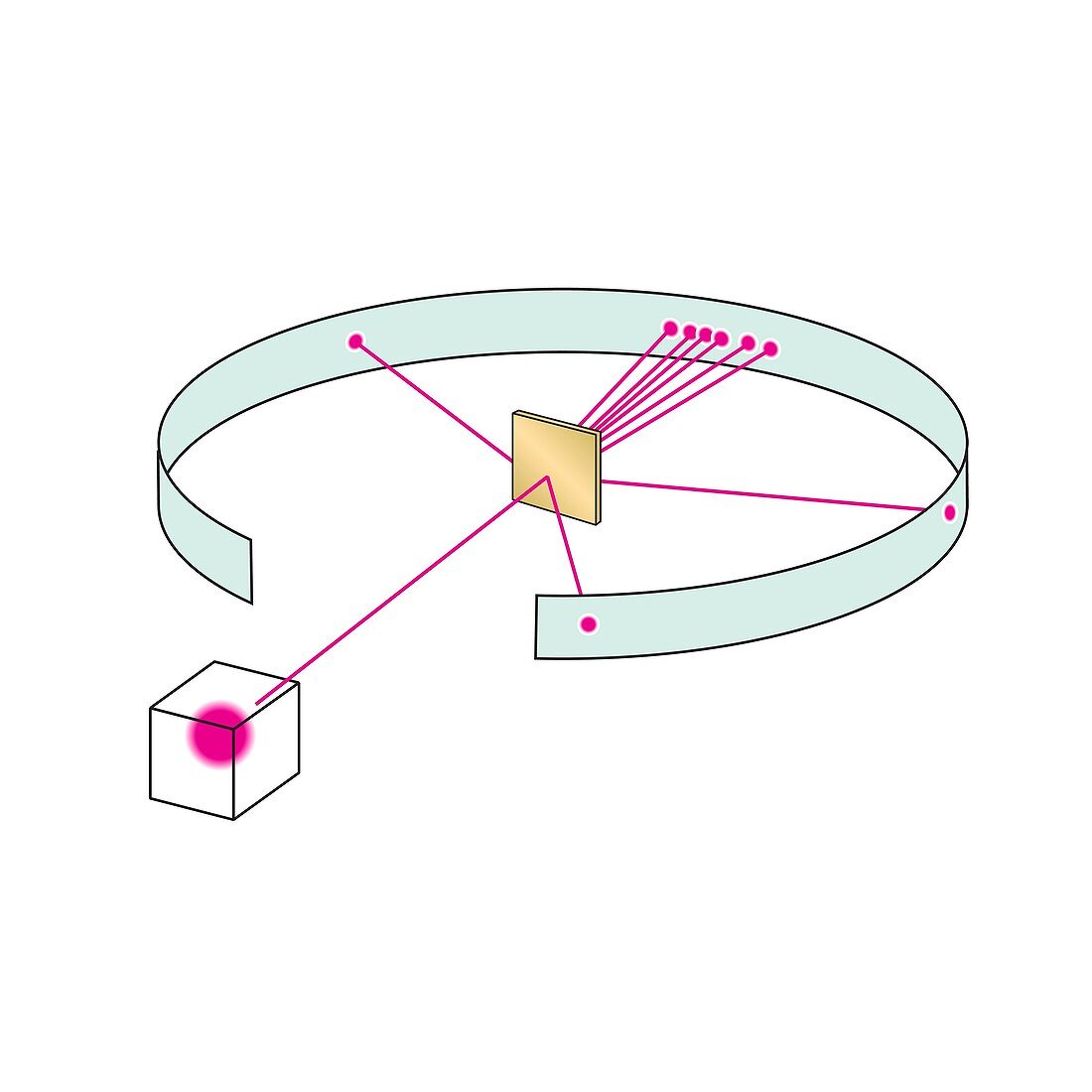
| Rutherford scattering experiment. Illustration of the apparatus used to show the elastic scattering of charged particles according to Coulomb's law, which describes the electrostatic interaction between electrically charged particles, as demonstrated by Ernest Rutherford in 1911. Helium ions (alpha particles from a radiation source, pink) are fired at thin gold foil (centre) and the scattering pattern is observed on the ring-shaped surface. Most particles are deflected by only a small amount, but a few are deflected by large amounts, implying that the atomic nucleus is very small and positively charged. This phenomenon led to the development of the planetary Rutherford model of the atom and eventually the Bohr model. | |
| Lizenzart: | Lizenzpflichtig |
| Credit: | Science Photo Library |
| Bildgröße: | 4181 px × 4181 px |
| Modell-Rechte: | nicht erforderlich |
| Eigentums-Rechte: | nicht erforderlich |
| Restrictions: | - |
Preise für dieses Bild ab 15 €
Universitäten & Organisationen
(Informationsmaterial Digital, Informationsmaterial Print, Lehrmaterial Digital etc.)
ab 15 €
Redaktionell
(Bücher, Bücher: Sach- und Fachliteratur, Digitale Medien (redaktionell) etc.)
ab 30 €
Werbung
(Anzeigen, Aussenwerbung, Digitale Medien, Fernsehwerbung, Karten, Werbemittel, Zeitschriften etc.)
ab 55 €
Handelsprodukte
(bedruckte Textilie, Kalender, Postkarte, Grußkarte, Verpackung etc.)
ab 75 €
Pauschalpreise
Rechtepakete für die unbeschränkte Bildnutzung in Print oder Online
ab 495 €
Keywords
- 1900er Jahre,
- 20. Jahrhundert,
- Atom,
- atomar,
- Atome,
- Atomkern,
- ausgeschnitten,
- Ausrüstung,
- Ausschnitte,
- Bildung,
- Bildungs-,
- Demonstration,
- demonstrierend,
- Diagramm,
- Elektrisch,
- Elektrizität,
- Energie,
- Ernest Rutherford,
- Experiment,
- experimentell,
- gcse,
- geladene Partikel,
- Gerät,
- Geschichte,
- Gold,
- Grafik,
- historisch,
- Illustration,
- Info-Grafiken,
- Infografik,
- Infografiken,
- Informationsgrafiken,
- Ionen,
- Kerne,
- Kunstwerk,
- Messung,
- Muster,
- Naturwissenschaft in der Schule,
- Niemand,
- nuklear,
- Partikel,
- Physik,
- physisch,
- positive Ladung,
- radioaktiv,
- Radioaktivität,
- Strahlung,
- subatomic,
- Verstreut,
- weißer Hintergrund,
- Wissenschaftspädagogik,
- Zeichnung
