Radioactive isotopes affecting the human body, illustration
Bildnummer 12947969
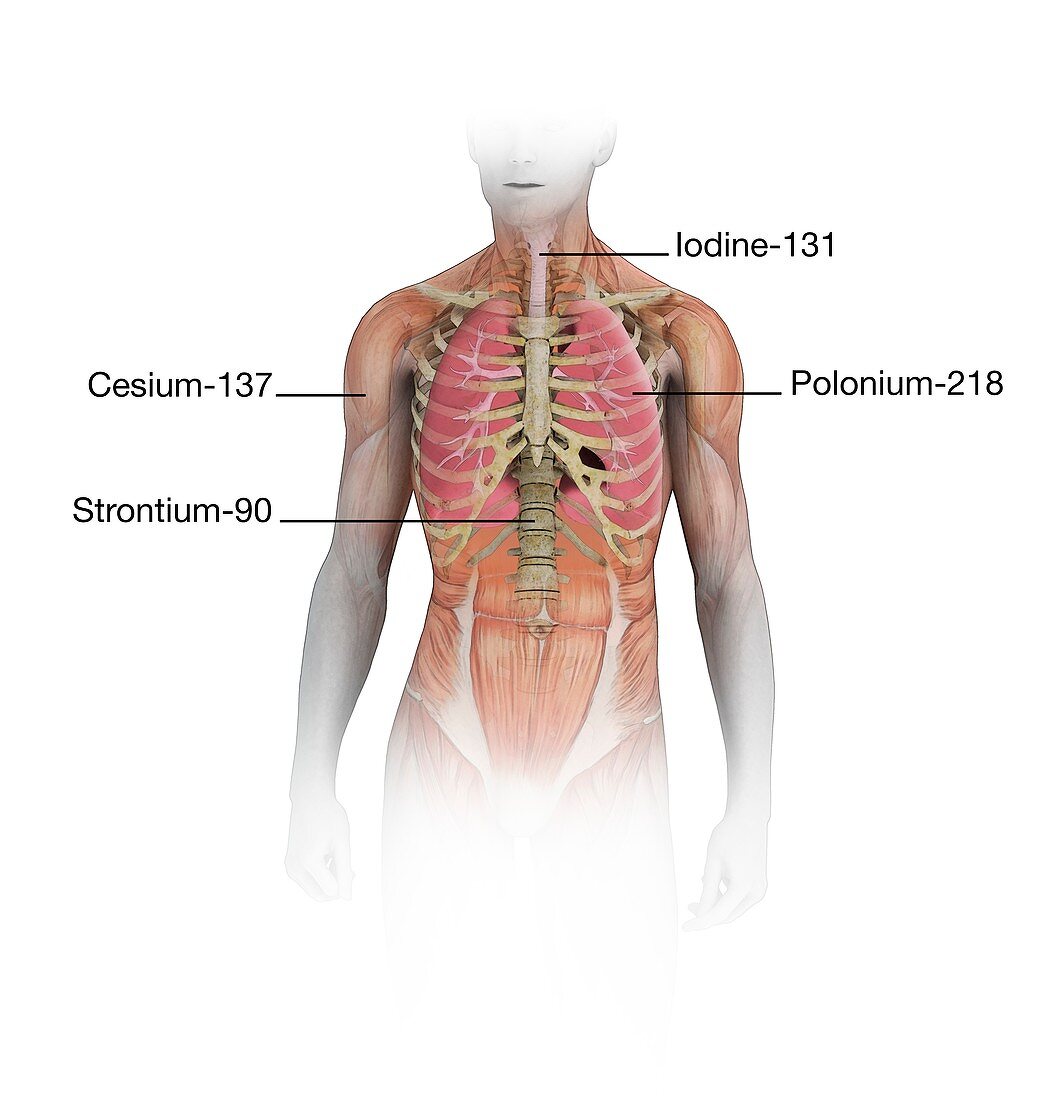
| Radioactive isotopes affecting the human body, illustration. Different isotopes have different effects as they penetrate the body to different depths. Caesium-137 is a by-product of nuclear fission in nuclear power stations and nuclear weapons. Similar to potassium, it forms water-soluble compounds and is concentrated most in soft tissues. It has a half-life of about 70 days. Strontium-90 is produced in nuclear fallout from weapons testing and accidents. It has a half-life of 28.9 years and can reach bones and cause bone cancer. Iodine-131 has a half-life of 8 days. It is also a major nuclear fission product. It becomes concentrated in thyroid tissues, where it may cause cancer. Polonium-218, with a half-life of just over three minutes, is part of the decay chain from radon, a radioactive gas that is breathed into the lungs. | |
| Lizenzart: | Lizenzpflichtig |
| Credit: | Science Photo Library / Jensen, Mikkel Juul |
| Bildgröße: | 4736 px × 4904 px |
| Modell-Rechte: | nicht erforderlich |
| Eigentums-Rechte: | nicht erforderlich |
| Restrictions: | - |
Preise für dieses Bild ab 15 €
Universitäten & Organisationen
(Informationsmaterial Digital, Informationsmaterial Print, Lehrmaterial Digital etc.)
ab 15 €
Redaktionell
(Bücher, Bücher: Sach- und Fachliteratur, Digitale Medien (redaktionell) etc.)
ab 30 €
Werbung
(Anzeigen, Aussenwerbung, Digitale Medien, Fernsehwerbung, Karten, Werbemittel, Zeitschriften etc.)
ab 55 €
Handelsprodukte
(bedruckte Textilie, Kalender, Postkarte, Grußkarte, Verpackung etc.)
ab 75 €
Pauschalpreise
Rechtepakete für die unbeschränkte Bildnutzung in Print oder Online
ab 495 €
Keywords
- abnormal,
- beschriftet,
- Cäsium-137,
- Etikette,
- Etiketten,
- geduldig,
- Illustration,
- Isotop,
- Jod,
- Knochen,
- Krebserregend,
- Kunstwerk,
- Lunge,
- Lungen,
- Lungenkrebs,
- Medizin,
- medizinisch,
- menschlicher Körper,
- Muskel,
- Niemand,
- Physik,
- physisch,
- Radioaktivität,
- Risiko,
- Rückgrat,
- sr,
- Strahlung,
- Text,
- Thyreoidea,
- ungesund,
- Verschmutzung,
- weißer Hintergrund,
- Wirbelsäule,
- Wirbelsäulen-
