Copper reactivity
Bildnummer 12653487
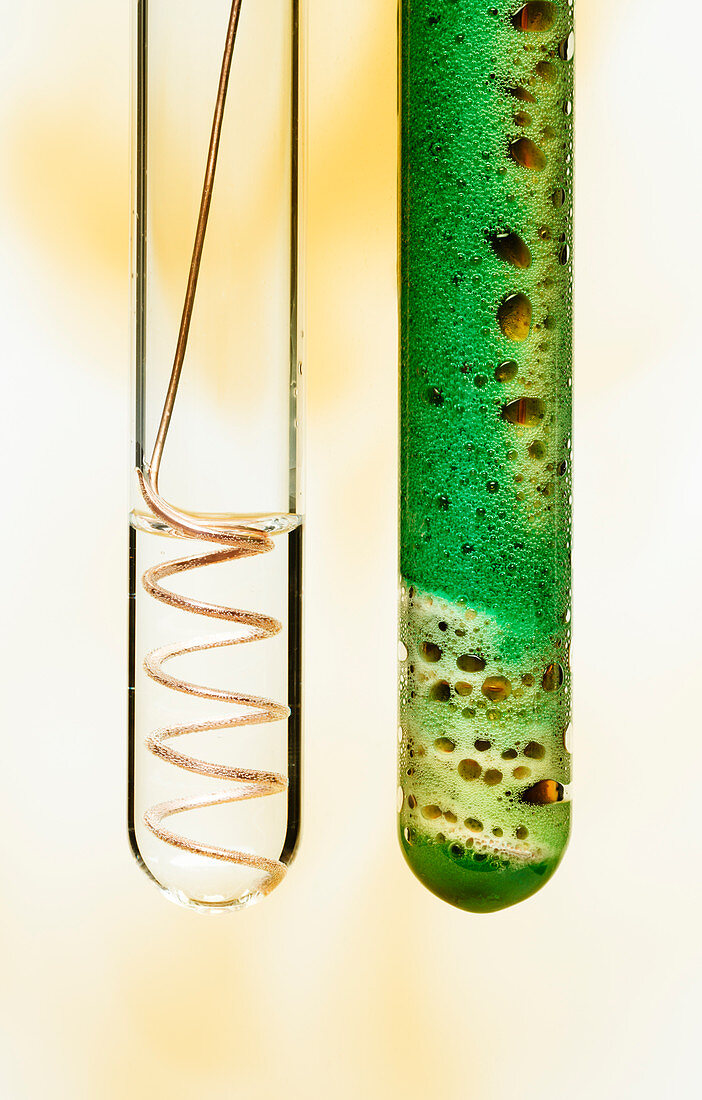
| Copper reactivity. Copper metal (Cu) coils are placed in test tubes with concentrated (17M, right) and diluted (1M, left) nitric acid (HNO3, right). Copper reacts vigorously with concentrated nitric acid and forms copper nitrate (Cu(NO3)2) and brown nitrogen dioxide gas (NO2) bubbles: Cu + HNO3 -> Cu(NO3)2 + NO2 + H2O. Copper reacts much slower with diluted nitric acid and forms copper nitrate (Cu(NO3)2) and colorless nitric oxide gas (NO) bubbles: Cu + HNO3 -> Cu(NO3)2 + NO + H2O. | |
| Lizenzart: | Lizenzpflichtig |
| Credit: | Science Photo Library / Science Source / Turtle Rock Scientific |
| Bildgröße: | 3280 px × 5139 px |
| Modell-Rechte: | nicht erforderlich |
| Eigentums-Rechte: | nicht erforderlich |
| Restrictions: | - |
Preise für dieses Bild ab 15 €
Universitäten & Organisationen
(Informationsmaterial Digital, Informationsmaterial Print, Lehrmaterial Digital etc.)
ab 15 €
Redaktionell
(Bücher, Bücher: Sach- und Fachliteratur, Digitale Medien (redaktionell) etc.)
ab 30 €
Werbung
(Anzeigen, Aussenwerbung, Digitale Medien, Fernsehwerbung, Karten, Werbemittel, Zeitschriften etc.)
ab 55 €
Handelsprodukte
(bedruckte Textilie, Kalender, Postkarte, Grußkarte, Verpackung etc.)
ab 75 €
Pauschalpreise
Rechtepakete für die unbeschränkte Bildnutzung in Print oder Online
ab 495 €
Keywords
- Acid,
- Chemie,
- chemisch,
- cu,
- Draht,
- elementar,
- Gas,
- HNO3,
- Konzentration,
- Konzentriert,
- Kupfer,
- Luftblasen,
- Metall,
- NO2,
- Oxidation,
- Reagenzglas,
- Reaktion,
- Reduzierung,
- Spule,
- Verdünnt,
- Vergleich
