Iron(II) hydroxide precipitate
Bildnummer 12647149
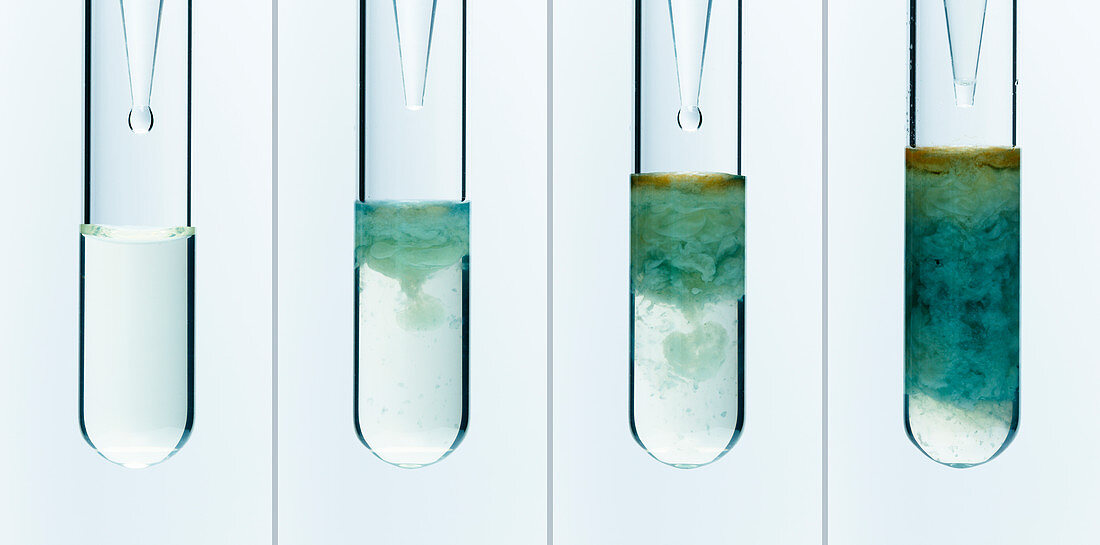
| Iron(II) hydroxide precipitate. Iron(II) hydroxide precipitate (Fe(OH)2) is formed when 1M solution of sodium hydroxide (NaOH) is added from a pipette to 0.2M solution of iron(II) ammonium sulfate (Fe(NH4)2(SO4)2) in a test tube. The reaction is Fe2+ + OH- -> Fe(OH)2. Pure iron(II) hydroxide is white, however even trace amounts of oxygen make it greenish. | |
| Lizenzart: | Lizenzpflichtig |
| Credit: | Science Photo Library / Science Source / Turtle Rock Scientific |
| Bildgröße: | 7100 px × 3515 px |
| Modell-Rechte: | nicht erforderlich |
| Eigentums-Rechte: | nicht erforderlich |
| Restrictions: | - |
Preise für dieses Bild ab 15 €
Universitäten & Organisationen
(Informationsmaterial Digital, Informationsmaterial Print, Lehrmaterial Digital etc.)
ab 15 €
Redaktionell
(Bücher, Bücher: Sach- und Fachliteratur, Digitale Medien (redaktionell) etc.)
ab 30 €
Werbung
(Anzeigen, Aussenwerbung, Digitale Medien, Fernsehwerbung, Karten, Werbemittel, Zeitschriften etc.)
ab 55 €
Handelsprodukte
(bedruckte Textilie, Kalender, Postkarte, Grußkarte, Verpackung etc.)
ab 75 €
Pauschalpreise
Rechtepakete für die unbeschränkte Bildnutzung in Print oder Online
ab 495 €
