Lead sulphate precipitate, 1 of 3
Bildnummer 12629943
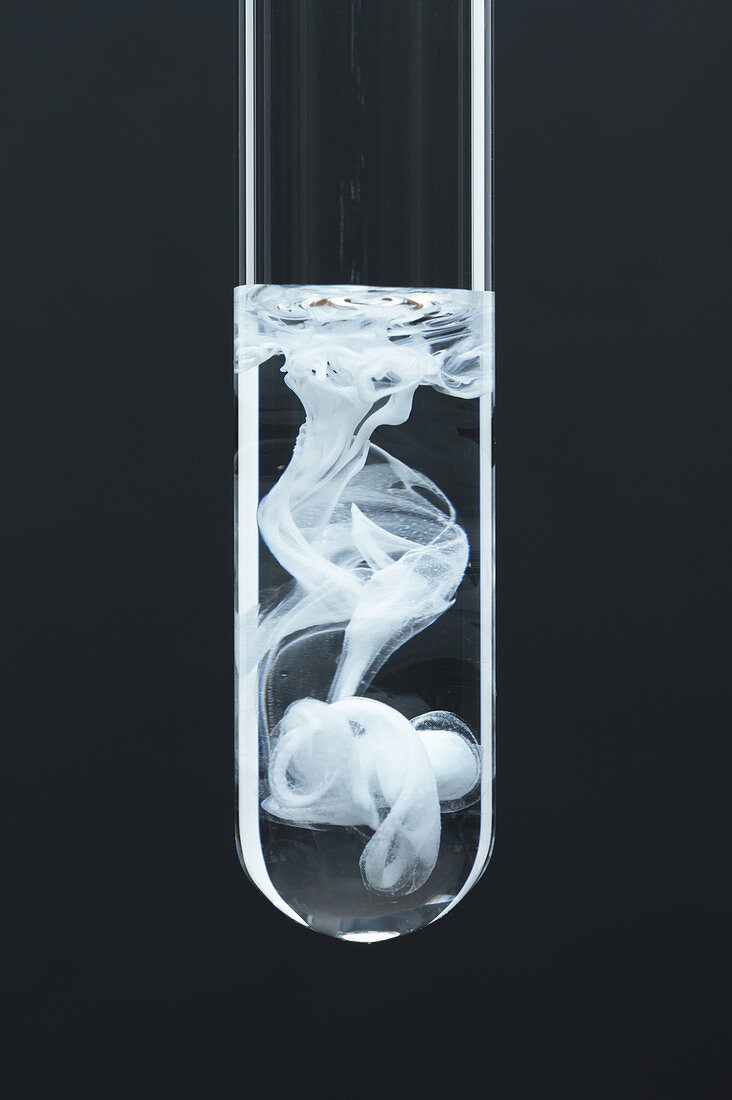
| Lead sulphate precipitate, 1 of 3. This series shows a progression as 0.5 M solution of copper sulphate (CuSO4) is added drop by drop to a test tube containing 0.2 M solution of lead (II) nitrate (Pb(NO3)2). Lead (II) sulphate precipitate (PbSO4) is formed as a result: CuSO4 + Pb(NO3)2 -> PbSO4 + Cu(NO3)2. This is an example of a double displacement reaction. | |
| Lizenzart: | Lizenzpflichtig |
| Credit: | Science Photo Library / Giphotostock |
| Bildgröße: | 3406 px × 5119 px |
| Modell-Rechte: | nicht erforderlich |
| Eigentums-Rechte: | nicht erforderlich |
| Restrictions: | - |
Preise für dieses Bild ab 15 €
Universitäten & Organisationen
(Informationsmaterial Digital, Informationsmaterial Print, Lehrmaterial Digital etc.)
ab 15 €
Redaktionell
(Bücher, Bücher: Sach- und Fachliteratur, Digitale Medien (redaktionell) etc.)
ab 30 €
Werbung
(Anzeigen, Aussenwerbung, Digitale Medien, Fernsehwerbung, Karten, Werbemittel, Zeitschriften etc.)
ab 55 €
Handelsprodukte
(bedruckte Textilie, Kalender, Postkarte, Grußkarte, Verpackung etc.)
ab 75 €
Pauschalpreise
Rechtepakete für die unbeschränkte Bildnutzung in Print oder Online
ab 495 €
