Isotopes of hydrogen,illustration
Bildnummer 11731261
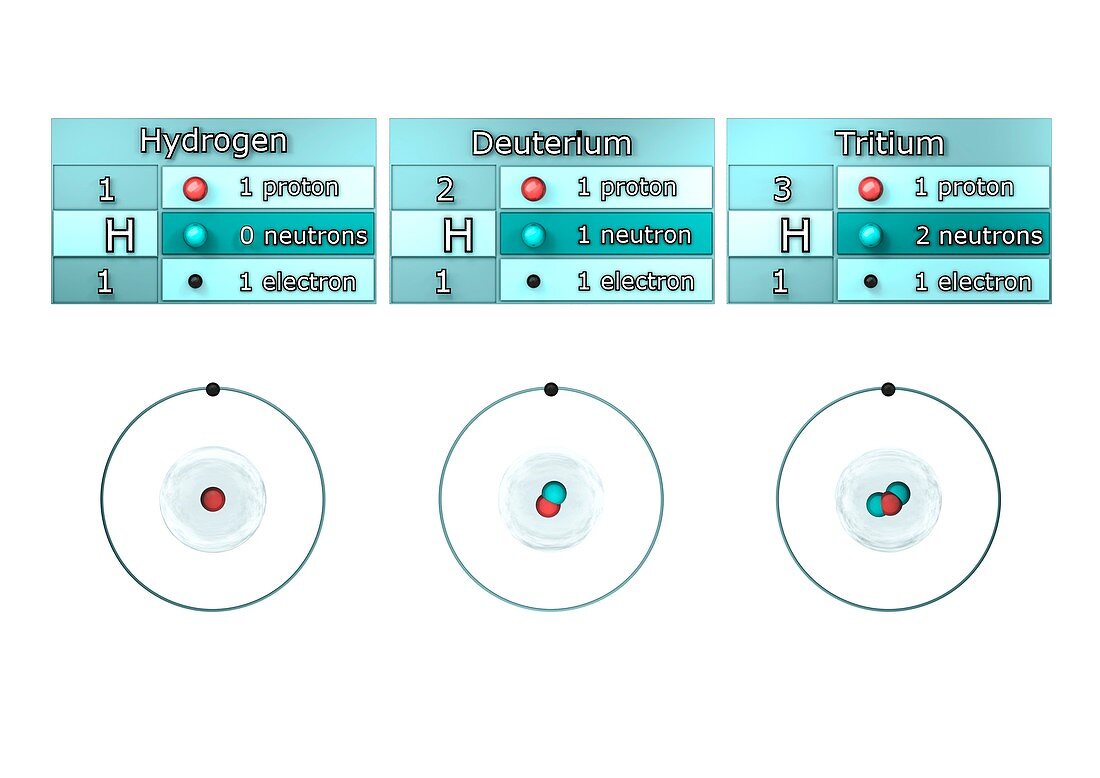
| Isotopes of hydrogen. Illustration showing the isotopes of hydrogen. Isotopes are forms of an element that contain different numbers of neutrons in the atomic nucleus. Hydrogen-1,known as protium,has one proton (red) and no neutrons in its nucleus,hydrogen-2,deuterium,has one proton and one neutron (blue),and hydrogen-3,tritium,has one proton and two neutrons . All the isotopes also have one electron (black) orbiting the nucleus. Tritium is a radioactive isotope,but protium and deuterium are stable | |
| Lizenzart: | Lizenzpflichtig |
| Credit: | Science Photo Library / Animate4.com |
| Bildgröße: | 4978 px × 3519 px |
| Modell-Rechte: | nicht erforderlich |
| Eigentums-Rechte: | nicht erforderlich |
| Restrictions: | - |
Preise für dieses Bild ab 15 €
Universitäten & Organisationen
(Informationsmaterial Digital, Informationsmaterial Print, Lehrmaterial Digital etc.)
ab 15 €
Redaktionell
(Bücher, Bücher: Sach- und Fachliteratur, Digitale Medien (redaktionell) etc.)
ab 30 €
Werbung
(Anzeigen, Aussenwerbung, Digitale Medien, Fernsehwerbung, Karten, Werbemittel, Zeitschriften etc.)
ab 55 €
Handelsprodukte
(bedruckte Textilie, Kalender, Postkarte, Grußkarte, Verpackung etc.)
ab 75 €
Pauschalpreise
Rechtepakete für die unbeschränkte Bildnutzung in Print oder Online
ab 495 €
Keywords
- Atom,
- atomar,
- atomare Struktur,
- Atomkern,
- Bohr,
- cgi,
- Chemie,
- chemisch,
- Darstellung,
- Diagramm,
- digital erzeugtes Bild,
- Drei,
- Elektron,
- Element,
- Illustration,
- Isotop,
- Kunstwerk,
- Modell-,
- Niemand,
- Orbit,
- Partikel,
- Physik,
- physisch,
- Protium,
- Proton,
- radioaktiv,
- Stabil,
- Struktur,
- Tisch,
- Trio,
- Vergleich,
- Wasserstoff,
- Wasserstoff-2,
- Wasserstoff-3,
- weißer Hintergrund
