States of matter,illustration
Bildnummer 11722184
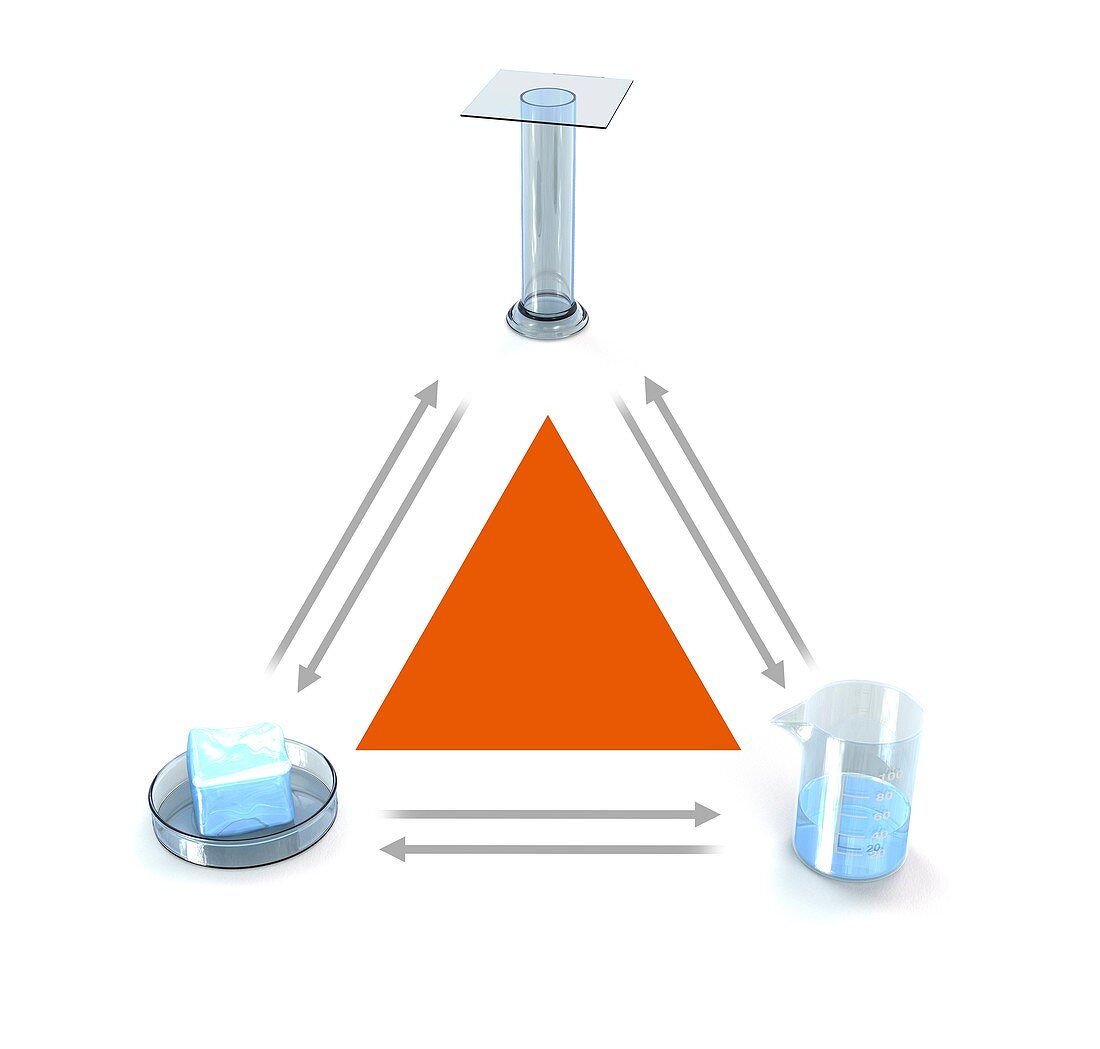
| States of matter. Computer illustration showing the three states of matter and how any one of them can be converted (arrows) directly into one of the other two - by an increase or reduction of energy in the form of heat. Clockwise from: gas,liquid and solid. Molecules are in constant motion. In solids,the molecules have a strong attraction,are close together and have limited motion. In a liquid,the molecules have more energy,which enables them to overcome some of the attraction and move more freely. In a gas,attraction between particles is minimised and they move freely throughout the container | |
| Lizenzart: | Lizenzpflichtig |
| Credit: | Science Photo Library / Jensen, Mikkel Juul |
| Bildgröße: | 5308 px × 5063 px |
| Modell-Rechte: | nicht erforderlich |
| Eigentums-Rechte: | nicht erforderlich |
| Restrictions: | - |
Preise für dieses Bild ab 15 €
Universitäten & Organisationen
(Informationsmaterial Digital, Informationsmaterial Print, Lehrmaterial Digital etc.)
ab 15 €
Redaktionell
(Bücher, Bücher: Sach- und Fachliteratur, Digitale Medien (redaktionell) etc.)
ab 30 €
Werbung
(Anzeigen, Aussenwerbung, Digitale Medien, Fernsehwerbung, Karten, Werbemittel, Zeitschriften etc.)
ab 55 €
Handelsprodukte
(bedruckte Textilie, Kalender, Postkarte, Grußkarte, Verpackung etc.)
ab 75 €
Pauschalpreise
Rechtepakete für die unbeschränkte Bildnutzung in Print oder Online
ab 495 €
Keywords
- ausgeschnitten,
- Ausschnitte,
- Becherglas,
- Chemie,
- chemisch,
- einfrieren,
- Energie,
- Flüssigkeit,
- Gas,
- Illustration,
- intermolekular,
- Kunstwerk,
- Macht,
- Molekül,
- Moleküle,
- Niemand,
- Partikel,
- Phase,
- Phasen,
- Schmelzen,
- solide,
- Staat,
- Struktur,
- Temperatur,
- Thermodynamik,
- Übergänge,
- Veränderung,
- weißer Hintergrund,
- Wissenschaftspädagogik
