Nickel,atomic structure
Bildnummer 11698424
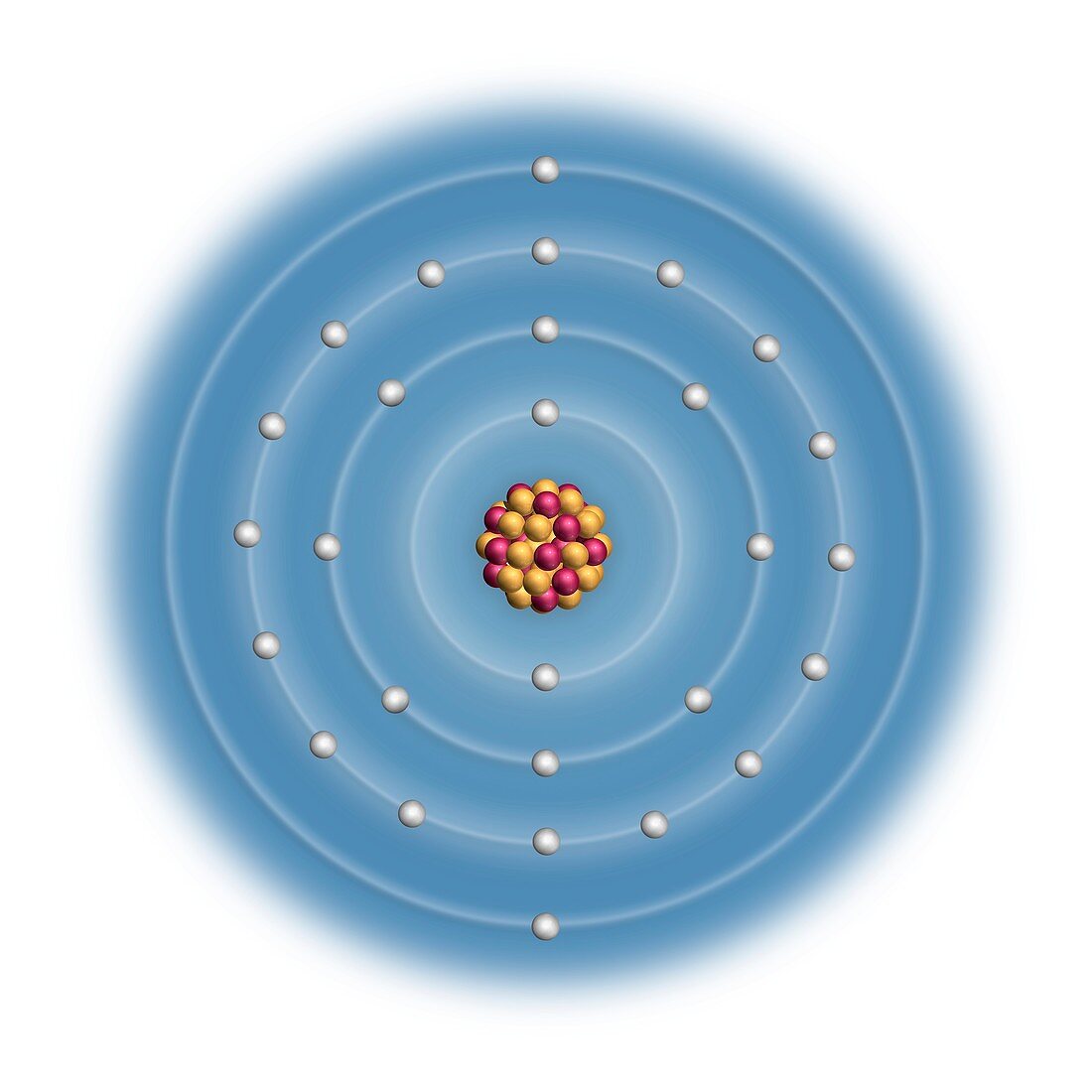
| Nickel (Ni). Diagram of the nuclear composition and electron configuration of an atom of nickel-58 (atomic number: 28),the most common isotope of this element. The nucleus consists of 28 protons (red) and 30 neutrons (yellow). 28 electrons (white) bind to the nucleus,successively occupying available electron shells (rings). Nickel is a transition metal in group 10,period 4,and the d-block of the periodic table. It has a melting point of 1455 degrees Celsius. The trends across the transition metals are due to electrons filling an inner d-subshell (here,within the 3rd ring),shielding the outer electrons from the increasing nuclear charge | |
| Lizenzart: | Lizenzpflichtig |
| Credit: | Science Photo Library / Clarivan, Carlos |
| Bildgröße: | 4180 px × 4180 px |
| Modell-Rechte: | nicht erforderlich |
| Eigentums-Rechte: | nicht erforderlich |
| Restrictions: | - |
Preise für dieses Bild ab 15 €
Universitäten & Organisationen
(Informationsmaterial Digital, Informationsmaterial Print, Lehrmaterial Digital etc.)
ab 15 €
Redaktionell
(Bücher, Bücher: Sach- und Fachliteratur, Digitale Medien (redaktionell) etc.)
ab 30 €
Werbung
(Anzeigen, Aussenwerbung, Digitale Medien, Fernsehwerbung, Karten, Werbemittel, Zeitschriften etc.)
ab 55 €
Handelsprodukte
(bedruckte Textilie, Kalender, Postkarte, Grußkarte, Verpackung etc.)
ab 75 €
Pauschalpreise
Rechtepakete für die unbeschränkte Bildnutzung in Print oder Online
ab 495 €
Keywords
- Atom,
- atomar,
- Atomkern,
- Chemie,
- chemische Eigenschaft,
- Chemische Eigenschaften,
- d-Block,
- Diagramm,
- Ebenen,
- einfache Elektronenkonfiguration,
- Elektronen,
- Elektronenhülle,
- Elektronenkonfiguration,
- elektronisch,
- Element,
- elementar,
- Elemente,
- Feststoffe,
- Illustration,
- Isotop,
- Metall,
- Metalle,
- Metallisch,
- Muscheln,
- Neutronen,
- ni,
- nuklear,
- Nukleon,
- Orbital,
- Orbitale,
- physikalische Chemie,
- physikalische Eigenschaft,
- physikalische Eigenschaften,
- Schale,
- solide,
- Struktur,
- strukturell,
- subatomic,
- Übergangsmetall,
- Übergangsmetalle,
- Unterschale,
- Unterschalen,
- weißer Hintergrund
