Francium,atomic structure
Bildnummer 11662062
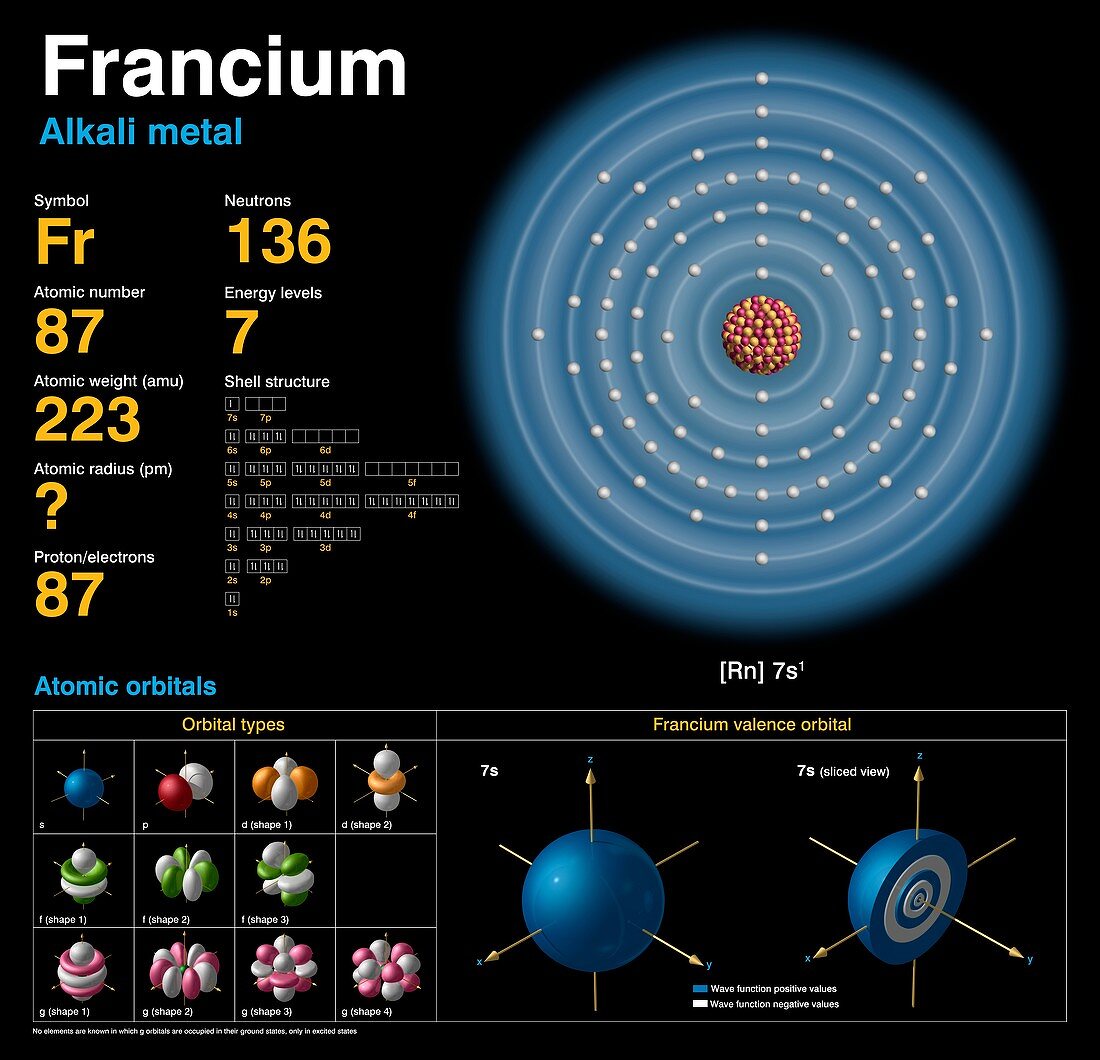
| Francium (Fr). Diagram of the nuclear composition,electron configuration,chemical data,and valence (outer electron) orbitals of an atom of francium-223 (atomic number: 87),the most stable isotope of this element. The nucleus consists of 87 protons (red) and 136 neutrons (orange). 87 electrons (white) successively occupy available electron shells (rings). Francium is a radioactive alkali metal in group 1,period 7,and the s-block of the periodic table. Named after France,it was discovered in 1939 by Marguerite Perey. Francium-223 forms in uranium and thorium ores. It has a half-life of 22 minutes,emitting a beta or alpha particle from its unstable nucleus | |
| Lizenzart: | Lizenzpflichtig |
| Credit: | Science Photo Library / Clarivan, Carlos |
| Bildgröße: | 5197 px × 5008 px |
| Modell-Rechte: | nicht erforderlich |
| Eigentums-Rechte: | nicht erforderlich |
| Restrictions: | - |
Preise für dieses Bild ab 15 €
Universitäten & Organisationen
(Informationsmaterial Digital, Informationsmaterial Print, Lehrmaterial Digital etc.)
ab 15 €
Redaktionell
(Bücher, Bücher: Sach- und Fachliteratur, Digitale Medien (redaktionell) etc.)
ab 30 €
Werbung
(Anzeigen, Aussenwerbung, Digitale Medien, Fernsehwerbung, Karten, Werbemittel, Zeitschriften etc.)
ab 55 €
Handelsprodukte
(bedruckte Textilie, Kalender, Postkarte, Grußkarte, Verpackung etc.)
ab 75 €
Pauschalpreise
Rechtepakete für die unbeschränkte Bildnutzung in Print oder Online
ab 495 €
Keywords
- 7s,
- Alkali Metalle,
- Alkalimetall,
- Atom,
- atomar,
- Atomkern,
- Chemie,
- chemische Daten,
- chemische Eigenschaft,
- Chemische Eigenschaften,
- Diagramm,
- Ebenen,
- einfache Elektronenkonfiguration,
- Elektronen,
- Elektronenhülle,
- Elektronenkonfiguration,
- elektronisch,
- Element,
- elementar,
- Elemente,
- Feststoffe,
- fr,
- Illustration,
- Isotop,
- Metall,
- Metalle,
- Metallisch,
- Muscheln,
- Neutronen,
- nuklear,
- Nukleon,
- Orbital,
- Orbitale,
- physikalische Chemie,
- physikalische Eigenschaft,
- physikalische Eigenschaften,
- radioaktiv,
- s-Block,
- Schale,
- schwarzer Hintergrund,
- solide,
- Struktur,
- strukturell,
- subatomic,
- Symbol,
- Unterschale,
- Unterschalen
