Boyle's law of gases,artwork
Bildnummer 11630040
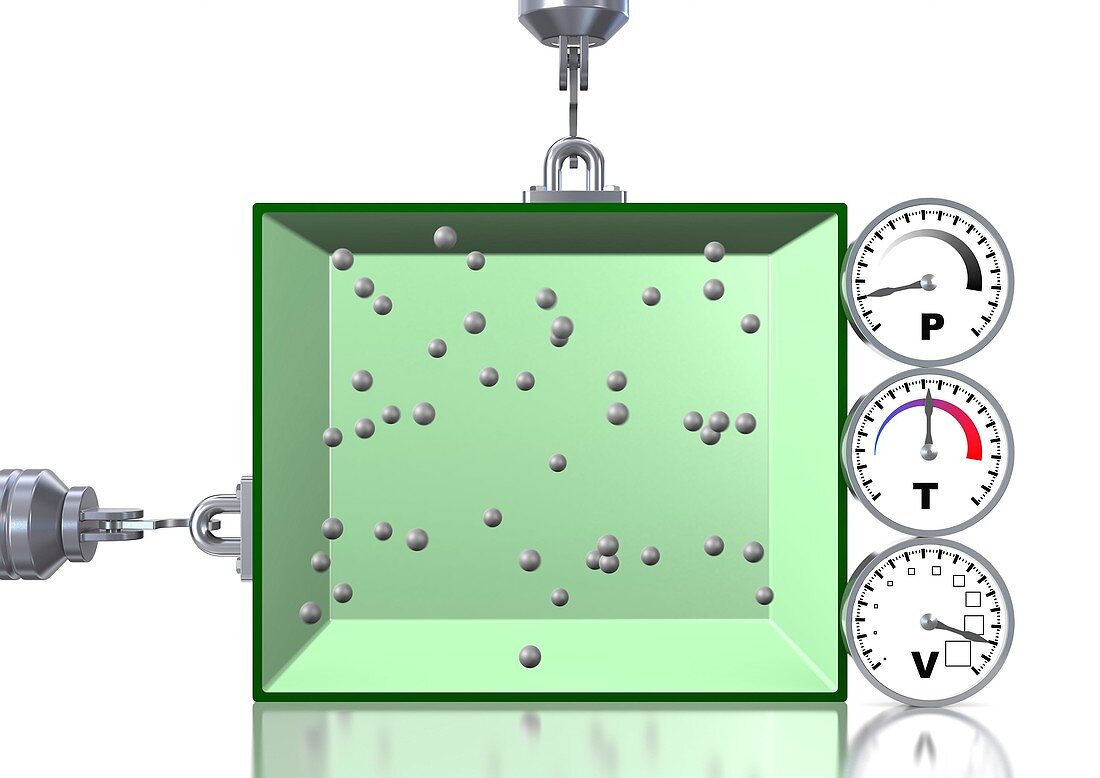
| Boyle's law of gases. Computer artwork explaining the gas laws as described by the combined gas law equation: PV=kT,which shows the relationship between the pressure (P),volume (V),and temperature (T) for a fixed number of gas molecules (spheres),where k is a constant. Here,the volume has been increased by enlarging the size of the container,while the temperature is kept constant. Therefore the pressure decreases; this is Boyle's law. The decrease in pressure is because the molecules have the same amount of energy but more space to move around,meaning they come into contact with the walls of the container less frequently | |
| Lizenzart: | Lizenzpflichtig |
| Credit: | Science Photo Library / Animated Healthcare |
| Bildgröße: | 5011 px × 3543 px |
| Modell-Rechte: | nicht erforderlich |
| Restrictions: | - |
Preise für dieses Bild ab 15 €
Universitäten & Organisationen
(Informationsmaterial Digital, Informationsmaterial Print, Lehrmaterial Digital etc.)
ab 15 €
Redaktionell
(Bücher, Bücher: Sach- und Fachliteratur, Digitale Medien (redaktionell) etc.)
ab 30 €
Werbung
(Anzeigen, Aussenwerbung, Digitale Medien, Fernsehwerbung, Karten, Werbemittel, Zeitschriften etc.)
ab 55 €
Handelsprodukte
(bedruckte Textilie, Kalender, Postkarte, Grußkarte, Verpackung etc.)
ab 75 €
Pauschalpreise
Rechtepakete für die unbeschränkte Bildnutzung in Print oder Online
ab 495 €
Keywords
- Atom,
- Atome,
- Avogadro-Gesetz,
- Bewegung,
- Box,
- Charles Gesetz,
- Container,
- Diagramm,
- Druck,
- Energie,
- Erklärung,
- Erläuterung,
- feste Masse,
- Gesetze,
- Groß,
- Illustration,
- Kinetik,
- kombiniert,
- Kunstwerk,
- Maß,
- Molekül,
- Moleküle,
- Physik,
- physisch,
- Skala,
- Temperatur,
- Thermodynamik,
- Volumen,
- weißer Hintergrund,
- Wissenschaftspädagogik,
- Zifferblätter
