'Kettle fur' limescale
Bildnummer 11520776
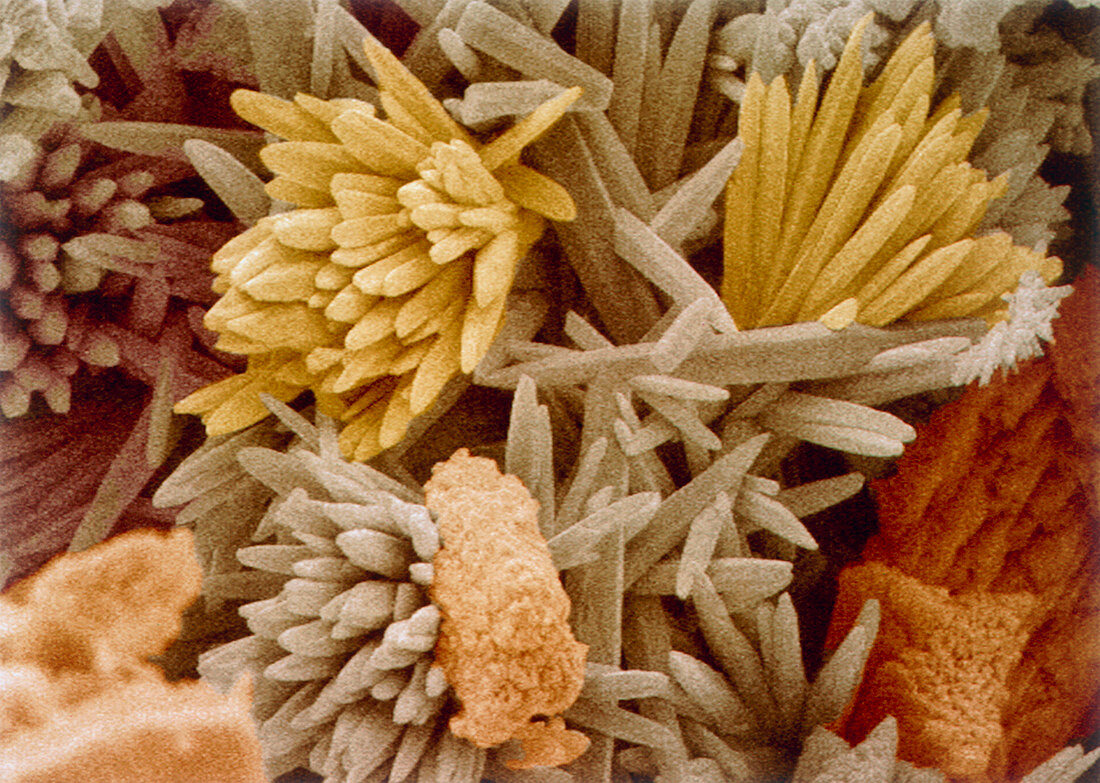
| Kettle fur. Coloured scanning electron micrograph (SEM) of fur from a domestic kettle,showing the crystals that form in a hard water area. The fur consists of crystals of calcium carbonate and crystals of calcium sulphate (needles). Hardness is caused by calcium salts; calcium bicarbonate (temporary) and calcium sulphate (permanent). When water containing calcium bicarbonate is boiled,the soluble bicarbonate breaks down to give the insoluble calcium carbonate. When water containing calcium sulphate is evaporated,boiled or dries out,the calcium sulphate precipitates out of solution. Magnification: x1,250 at 6x7cm size. x2,960 at 8x6",x1570 at 10x7cm master size | |
| Lizenzart: | Lizenzpflichtig |
| Credit: | Science Photo Library / Power And Syred |
| Bildgröße: | 5184 px × 3688 px |
| Modell-Rechte: | nicht erforderlich |
| Eigentums-Rechte: | nicht erforderlich |
| Restrictions: | - |
Preise für dieses Bild ab 15 €
Universitäten & Organisationen
(Informationsmaterial Digital, Informationsmaterial Print, Lehrmaterial Digital etc.)
ab 15 €
Redaktionell
(Bücher, Bücher: Sach- und Fachliteratur, Digitale Medien (redaktionell) etc.)
ab 30 €
Werbung
(Anzeigen, Aussenwerbung, Digitale Medien, Fernsehwerbung, Karten, Werbemittel, Zeitschriften etc.)
ab 55 €
Handelsprodukte
(bedruckte Textilie, Kalender, Postkarte, Grußkarte, Verpackung etc.)
ab 75 €
Pauschalpreise
Rechtepakete für die unbeschränkte Bildnutzung in Print oder Online
ab 495 €
