Artwork showing electron clouds of atoms
Bildnummer 11518604
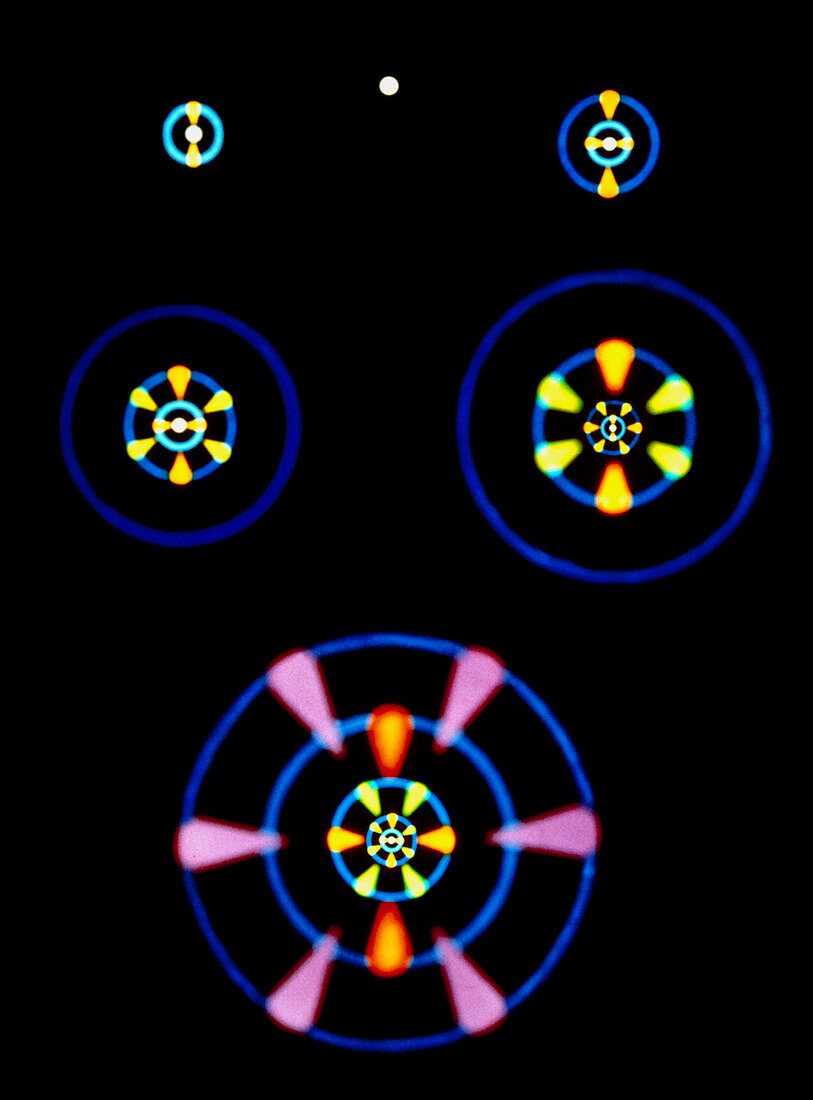
| Artwork showing the electron clouds,or orbitals,of atoms of hydrogen (top),carbon (top left),silicon (top right),iron (centre left),silver (centre right),& europium (bottom). As the number of electrons increases,additional orbitals are filled according to a regular pattern of spherical 's' orbitals (white to blue),dumbbell-shaped 'p' orbitals (orange),'d' orbitals (yellow-green) shaped like crossed dumbbells,& 'f' orbitals (purple). 'S' orbitals can contain up to 2 electrons,'p' orbitals up to 6,'d' orbitals up to 10,& 'f' orbitals up to 14. The extent to which an element's electrons fill these orbitals determines its chemical characteristics | |
| Lizenzart: | Lizenzpflichtig |
| Credit: | Science Photo Library / Parker, David |
| Bildgröße: | 2800 px × 3790 px |
| Modell-Rechte: | nicht erforderlich |
| Eigentums-Rechte: | nicht erforderlich |
| Restrictions: | - |
Preise für dieses Bild ab 15 €
Universitäten & Organisationen
(Informationsmaterial Digital, Informationsmaterial Print, Lehrmaterial Digital etc.)
ab 15 €
Redaktionell
(Bücher, Bücher: Sach- und Fachliteratur, Digitale Medien (redaktionell) etc.)
ab 30 €
Werbung
(Anzeigen, Aussenwerbung, Digitale Medien, Fernsehwerbung, Karten, Werbemittel, Zeitschriften etc.)
ab 55 €
Handelsprodukte
(bedruckte Textilie, Kalender, Postkarte, Grußkarte, Verpackung etc.)
ab 75 €
Pauschalpreise
Rechtepakete für die unbeschränkte Bildnutzung in Print oder Online
ab 495 €
