Boyle's law apparatus, illustration
Bildnummer 13376845
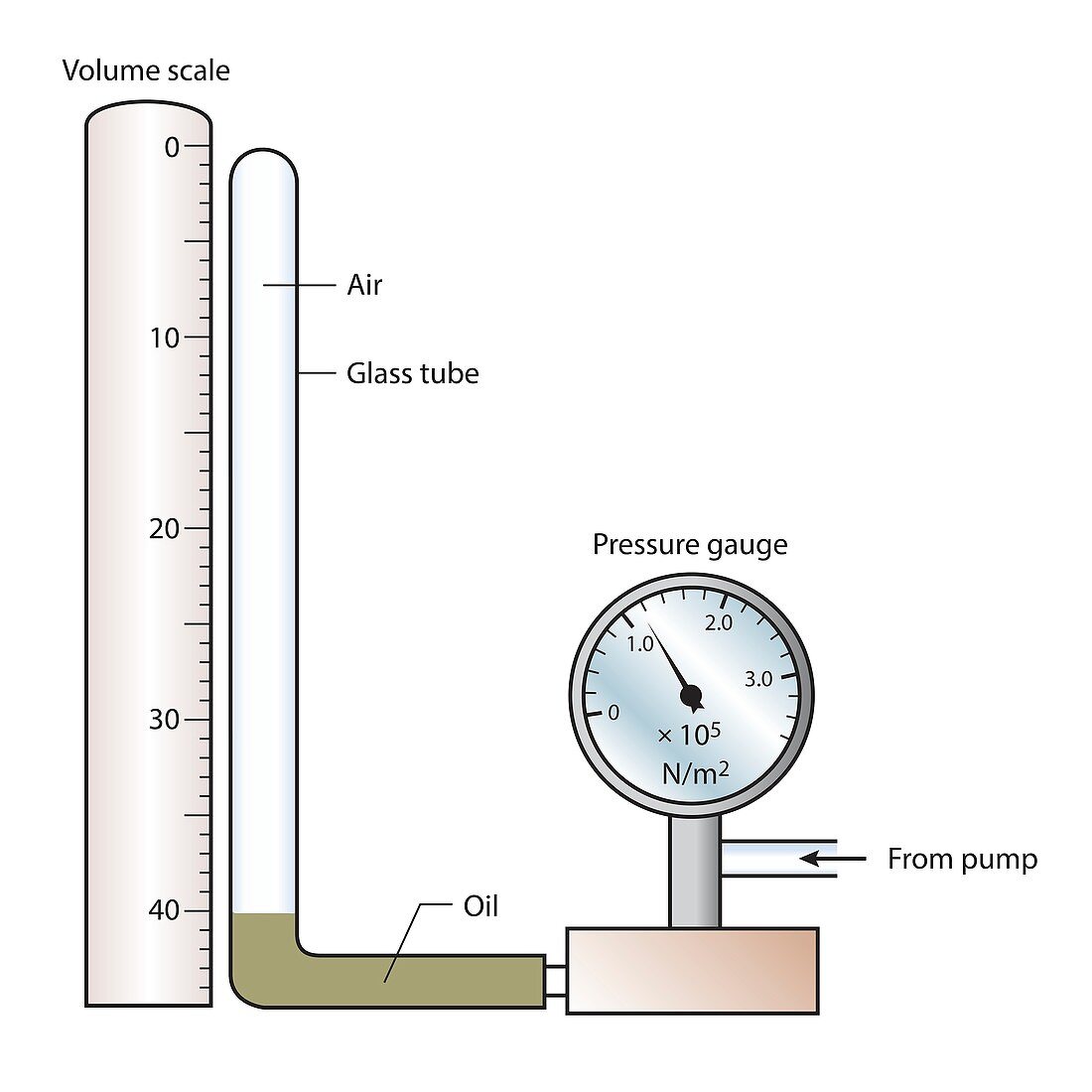
| Boyle's law of gases. Illustration showing apparatus used to demonstrate Boyle's law: in a closed system, the absolute pressure exerted by a given mass of an ideal gas is inversely proportional to the volume it occupies if the temperature and amount of gas remain unchanged. In this demonstration, if the pressure is increased using the pump, the oil is pushed up the glass tube and the volume of the air column decreases, as indicated by the scale on the left. | |
| Lizenzart: | Lizenzpflichtig |
| Credit: | Science Photo Library |
| Bildgröße: | 4181 px × 4181 px |
| Modell-Rechte: | nicht erforderlich |
| Eigentums-Rechte: | nicht erforderlich |
| Restrictions: | - |
Preise für dieses Bild ab 15 €
Universitäten & Organisationen
(Informationsmaterial Digital, Informationsmaterial Print, Lehrmaterial Digital etc.)
ab 15 €
Redaktionell
(Bücher, Bücher: Sach- und Fachliteratur, Digitale Medien (redaktionell) etc.)
ab 30 €
Werbung
(Anzeigen, Aussenwerbung, Digitale Medien, Fernsehwerbung, Karten, Werbemittel, Zeitschriften etc.)
ab 55 €
Handelsprodukte
(bedruckte Textilie, Kalender, Postkarte, Grußkarte, Verpackung etc.)
ab 75 €
Pauschalpreise
Rechtepakete für die unbeschränkte Bildnutzung in Print oder Online
ab 495 €
Keywords
- ausgeschnitten,
- Ausrüstung,
- Ausschnitte,
- beschriftet,
- Bildung,
- Bildungs-,
- Chemie,
- chemisch,
- Demonstration,
- demonstrierend,
- Diagramm,
- Druck,
- Energie,
- Etikette,
- Etiketten,
- Experiment,
- experimentell,
- feste Masse,
- Gas,
- gcse,
- Gerät,
- Hitze,
- Illustration,
- Info-Grafiken,
- Infografik,
- Infografiken,
- Informationsgrafiken,
- Kunstwerk,
- Labor,
- Maßstab,
- Messung,
- Naturwissenschaft in der Schule,
- Niemand,
- Physik,
- physisch,
- Pumpe,
- Recht,
- Robert Boyle,
- Tube,
- Variation,
- Veränderung,
- Volumen,
- weißer Hintergrund,
- Wissenschaftspädagogik,
- Zeichnung
