Iron, atomic structure
Bildnummer 12656184
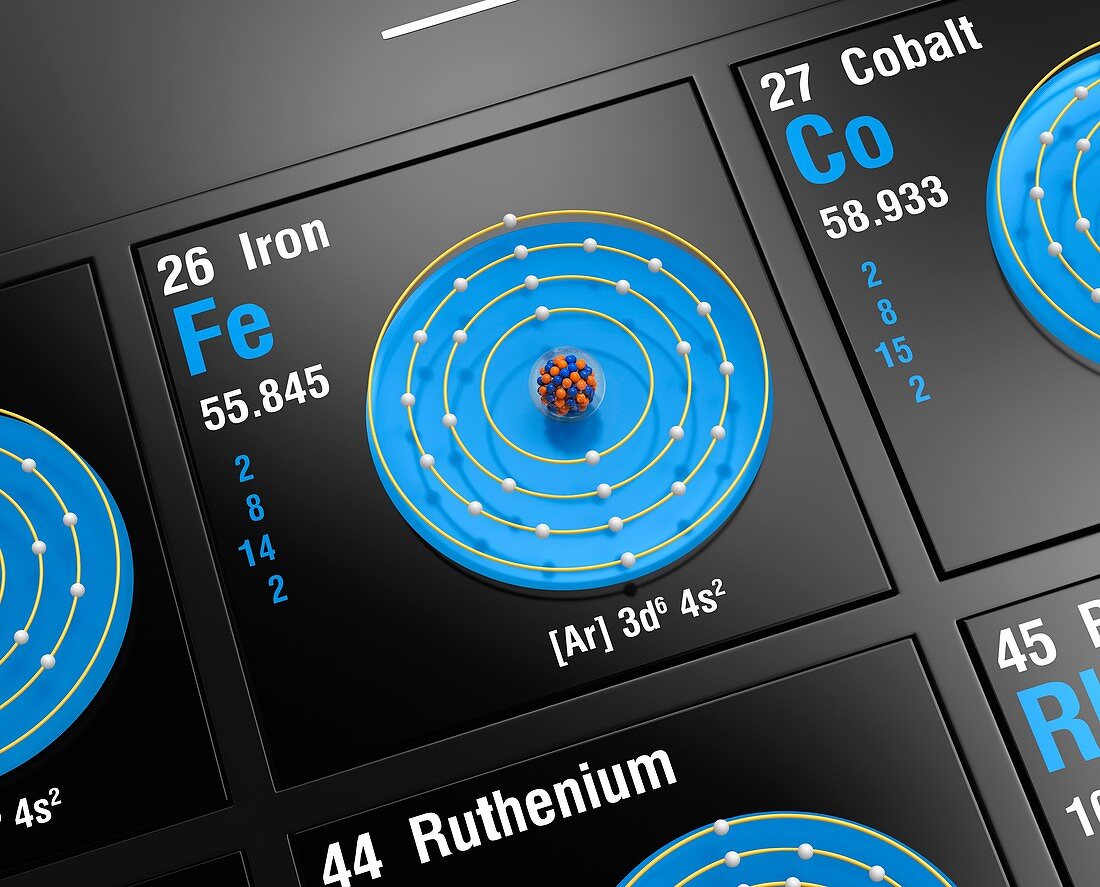
| Diagram of the nuclear composition, electron configuration, and valence orbitals of an atom of iron-56 (atomic number: 26), the most common isotope of this element. The nucleus consists of 26 protons (blue) and 30 neutrons (red). Iron is a transition metal in group 8, period 4, and the d-block of the periodic table. It has a melting point of 1538 degrees Celsius. The trends across the transition metals are due to electrons filling an inner d-subshell (here, within the 3rd ring), shielding the outer (valence) electrons from the increasing nuclear charge. | |
| Lizenzart: | Lizenzpflichtig |
| Credit: | Science Photo Library / Clarivan, Carlos |
| Bildgröße: | 4800 px × 3872 px |
| Modell-Rechte: | nicht erforderlich |
| Eigentums-Rechte: | nicht erforderlich |
| Restrictions: | - |
Preise für dieses Bild ab 15 €
Universitäten & Organisationen
(Informationsmaterial Digital, Informationsmaterial Print, Lehrmaterial Digital etc.)
ab 15 €
Redaktionell
(Bücher, Bücher: Sach- und Fachliteratur, Digitale Medien (redaktionell) etc.)
ab 30 €
Werbung
(Anzeigen, Aussenwerbung, Digitale Medien, Fernsehwerbung, Karten, Werbemittel, Zeitschriften etc.)
ab 55 €
Handelsprodukte
(bedruckte Textilie, Kalender, Postkarte, Grußkarte, Verpackung etc.)
ab 75 €
Pauschalpreise
Rechtepakete für die unbeschränkte Bildnutzung in Print oder Online
ab 495 €
Keywords
- Atom,
- atomar,
- Atomkern,
- Chemie,
- chemisch,
- chemische Eigenschaft,
- Chemische Eigenschaften,
- d-Block,
- Diagramm,
- Ebenen,
- einfache Elektronenkonfiguration,
- Eisen,
- Elektronen,
- Elektronenhülle,
- Elektronenkonfiguration,
- elektronisch,
- Element,
- elementar,
- Elemente,
- Feststoffe,
- Illustration,
- Isotop,
- Kunstwerk,
- Metall,
- Metalle,
- Metallisch,
- Muscheln,
- Neutronen,
- nuklear,
- Nukleon,
- Orbital,
- Orbitale,
- Periodensystem,
- physikalische Chemie,
- physikalische Eigenschaft,
- physikalische Eigenschaften,
- Schale,
- schwarzer Hintergrund,
- solide,
- Struktur,
- strukturell,
- subatomic,
- Übergangsmetall,
- Übergangsmetalle,
- Unterschale,
- Unterschalen,
- Volant
