Ammonium chloride formation
Bildnummer 13361098
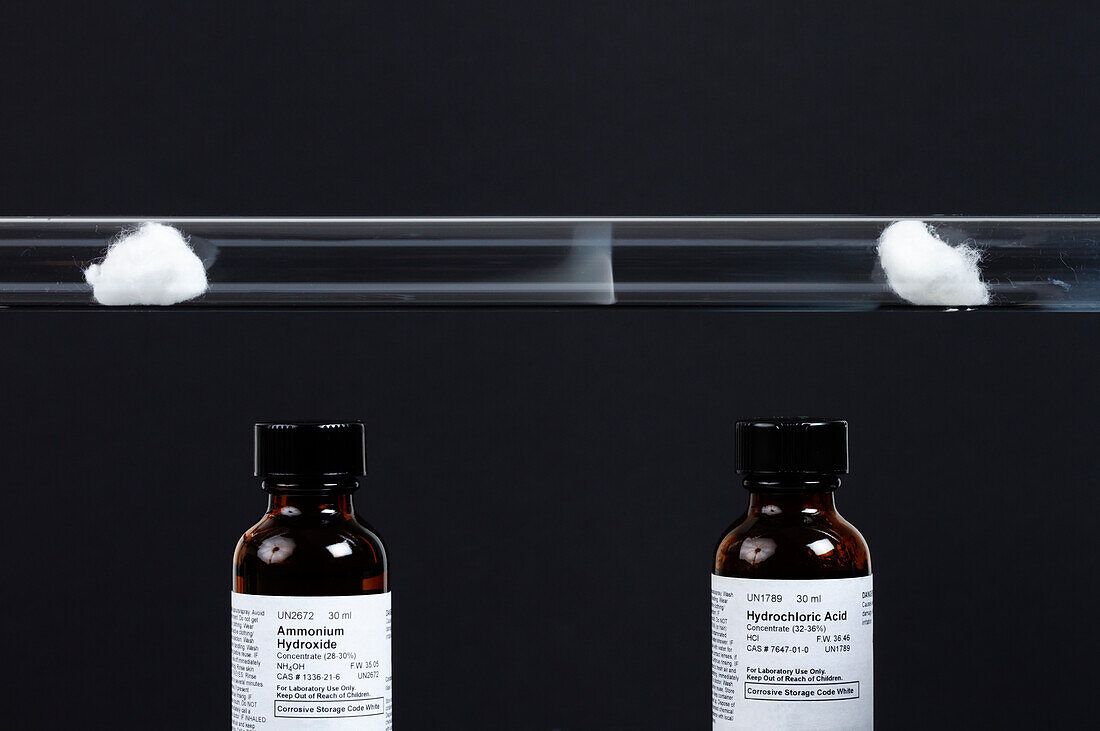
| Ammonium chloride formation. A glass tube is plugged by cotton balls at both ends. Small amounts of concentrated ammonium hydroxide (NH4OH, 28-30%) and concentrated hydrochloric acid (HCl, 32-36%) are used to wet the two balls. Colorless ammonia (NH3) and hydrogen chloride (HCl) gases diffuse from the respective balls. When they meet, they react and produce a ring of fine white powder of ammonium chloride: NH3 + HCl -> NH4Cl. Molecular weight of ammonia is less than that of hydrogen chloride, therefore it diffuses faster and the white ring is formed closer to the ball with the hydrochloric acid. | |
| Lizenzart: | Lizenzpflichtig |
| Credit: | Science Photo Library / Turtle Rock Scientific |
| Bildgröße: | 5160 px × 3429 px |
| Modell-Rechte: | nicht erforderlich |
| Eigentums-Rechte: | nicht erforderlich |
| Restrictions: | - |
Preise für dieses Bild ab 15 €
Universitäten & Organisationen
(Informationsmaterial Digital, Informationsmaterial Print, Lehrmaterial Digital etc.)
ab 15 €
Redaktionell
(Bücher, Bücher: Sach- und Fachliteratur, Digitale Medien (redaktionell) etc.)
ab 30 €
Werbung
(Anzeigen, Aussenwerbung, Digitale Medien, Fernsehwerbung, Karten, Werbemittel, Zeitschriften etc.)
ab 55 €
Handelsprodukte
(bedruckte Textilie, Kalender, Postkarte, Grußkarte, Verpackung etc.)
ab 75 €
Pauschalpreise
Rechtepakete für die unbeschränkte Bildnutzung in Print oder Online
ab 495 €
