Gas diffusion, illustration
Bildnummer 12969936
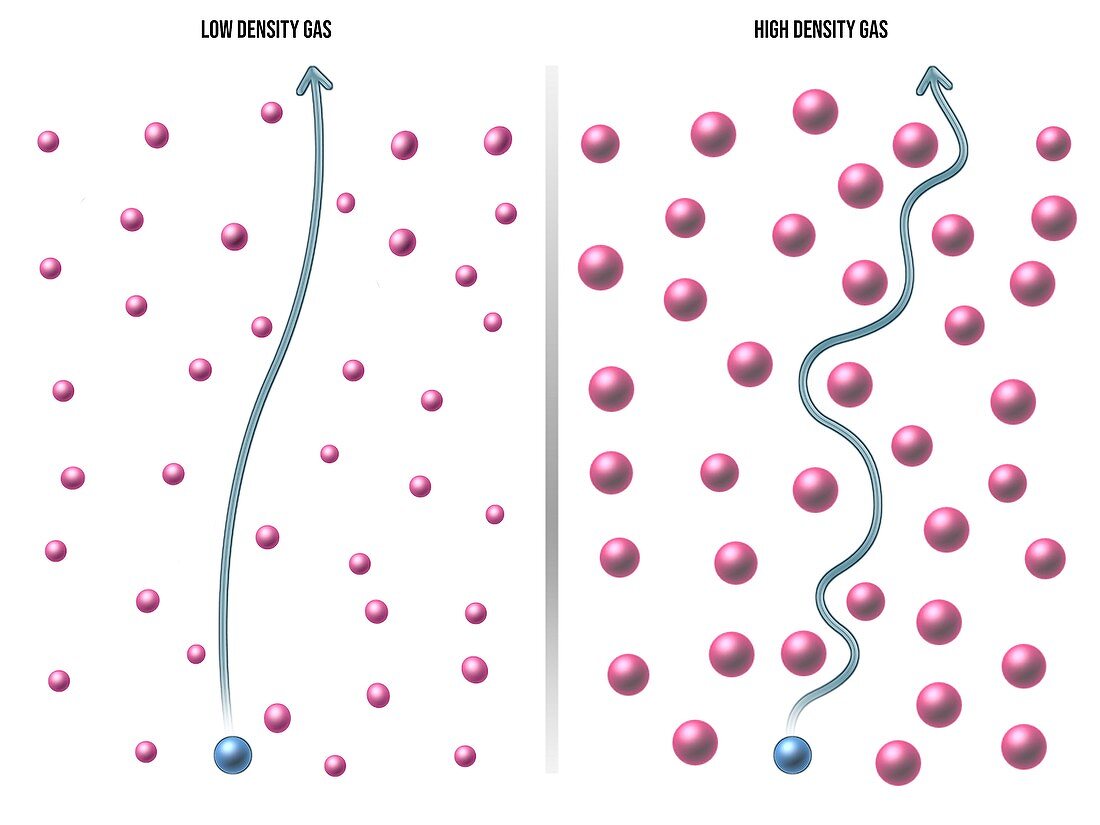
| Gas diffusion, illustration. Because gas molecules can move independently of each other and do so randomly, a gas spreads out from its source in a process called diffusion. The rate of diffusion depends on the density of the surrounding gas, as shown here by the path of a gas molecule through a low-density gas (left) and a high-density gas (right). | |
| Lizenzart: | Lizenzpflichtig |
| Credit: | Science Photo Library / De Angelis, Maurizio |
| Bildgröße: | 4827 px × 3620 px |
| Modell-Rechte: | nicht erforderlich |
| Eigentums-Rechte: | nicht erforderlich |
| Restrictions: | - |
Preise für dieses Bild ab 15 €
Universitäten & Organisationen
(Informationsmaterial Digital, Informationsmaterial Print, Lehrmaterial Digital etc.)
ab 15 €
Redaktionell
(Bücher, Bücher: Sach- und Fachliteratur, Digitale Medien (redaktionell) etc.)
ab 30 €
Werbung
(Anzeigen, Aussenwerbung, Digitale Medien, Fernsehwerbung, Karten, Werbemittel, Zeitschriften etc.)
ab 55 €
Handelsprodukte
(bedruckte Textilie, Kalender, Postkarte, Grußkarte, Verpackung etc.)
ab 75 €
Pauschalpreise
Rechtepakete für die unbeschränkte Bildnutzung in Print oder Online
ab 495 €
