Radon decaying to polonium in humans, illustration
Bildnummer 12947965
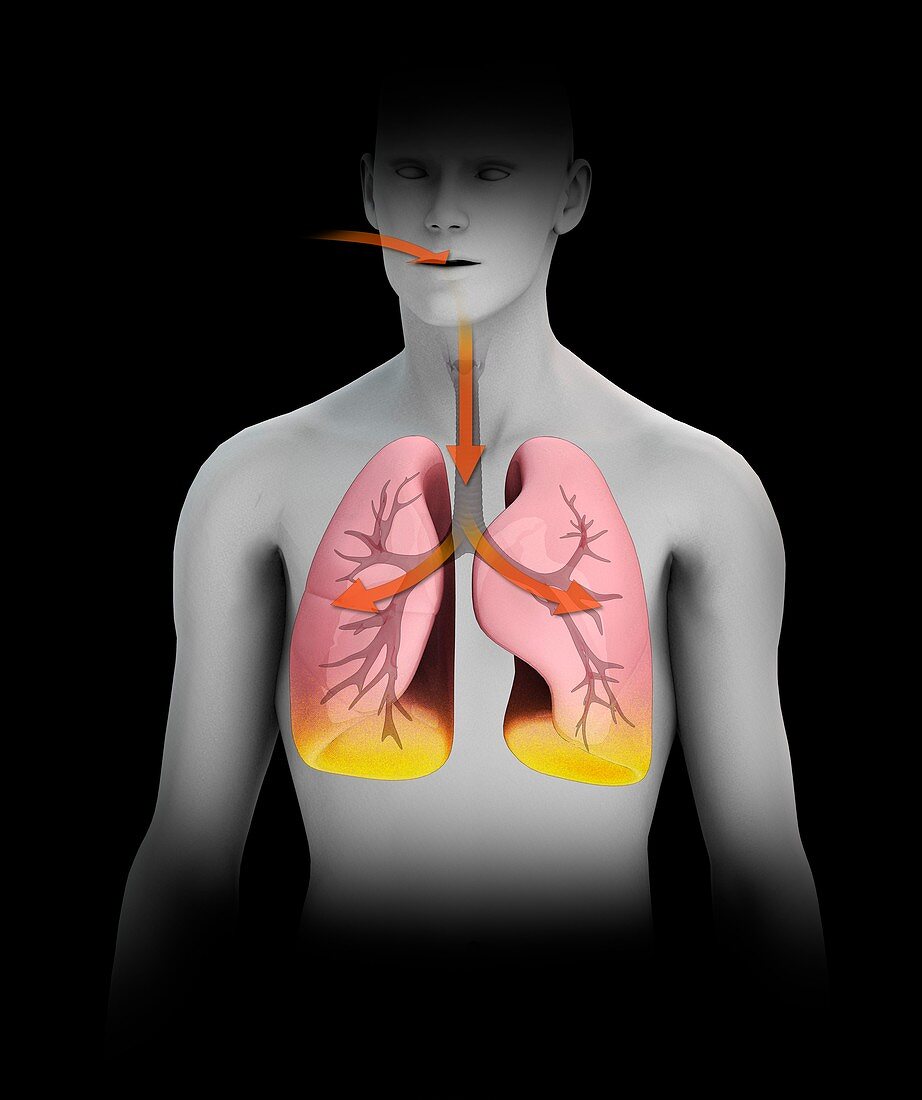
| Radon decaying to polonium in humans, illustration. Radon is a chemical element that is a radioactive gas that occurs in nature. It is being continuously generated from radioactive isotopes in the Earth's crust. The most stable isotope of radon is radon-222, which has a half-life of 3.8 days. As a gas, it can be inhaled into the lungs where it decays via alpha particle production to produce polonium-218, a solid that has a half-life of just over three minutes. Further decay takes places, eventually producing a stable isotope of lead. The effects of this can lead to an increased risk of lung cancer. For this illustration with labels, see image C047/4523. | |
| Lizenzart: | Lizenzpflichtig |
| Credit: | Science Photo Library / Jensen, Mikkel Juul |
| Bildgröße: | 5414 px × 6456 px |
| Modell-Rechte: | nicht erforderlich |
| Eigentums-Rechte: | nicht erforderlich |
| Restrictions: | - |
Preise für dieses Bild ab 15 €
Universitäten & Organisationen
(Informationsmaterial Digital, Informationsmaterial Print, Lehrmaterial Digital etc.)
ab 15 €
Redaktionell
(Bücher, Bücher: Sach- und Fachliteratur, Digitale Medien (redaktionell) etc.)
ab 30 €
Werbung
(Anzeigen, Aussenwerbung, Digitale Medien, Fernsehwerbung, Karten, Werbemittel, Zeitschriften etc.)
ab 55 €
Handelsprodukte
(bedruckte Textilie, Kalender, Postkarte, Grußkarte, Verpackung etc.)
ab 75 €
Pauschalpreise
Rechtepakete für die unbeschränkte Bildnutzung in Print oder Online
ab 495 €
