Calcium reacting with water, initial stage
Bildnummer 12644199
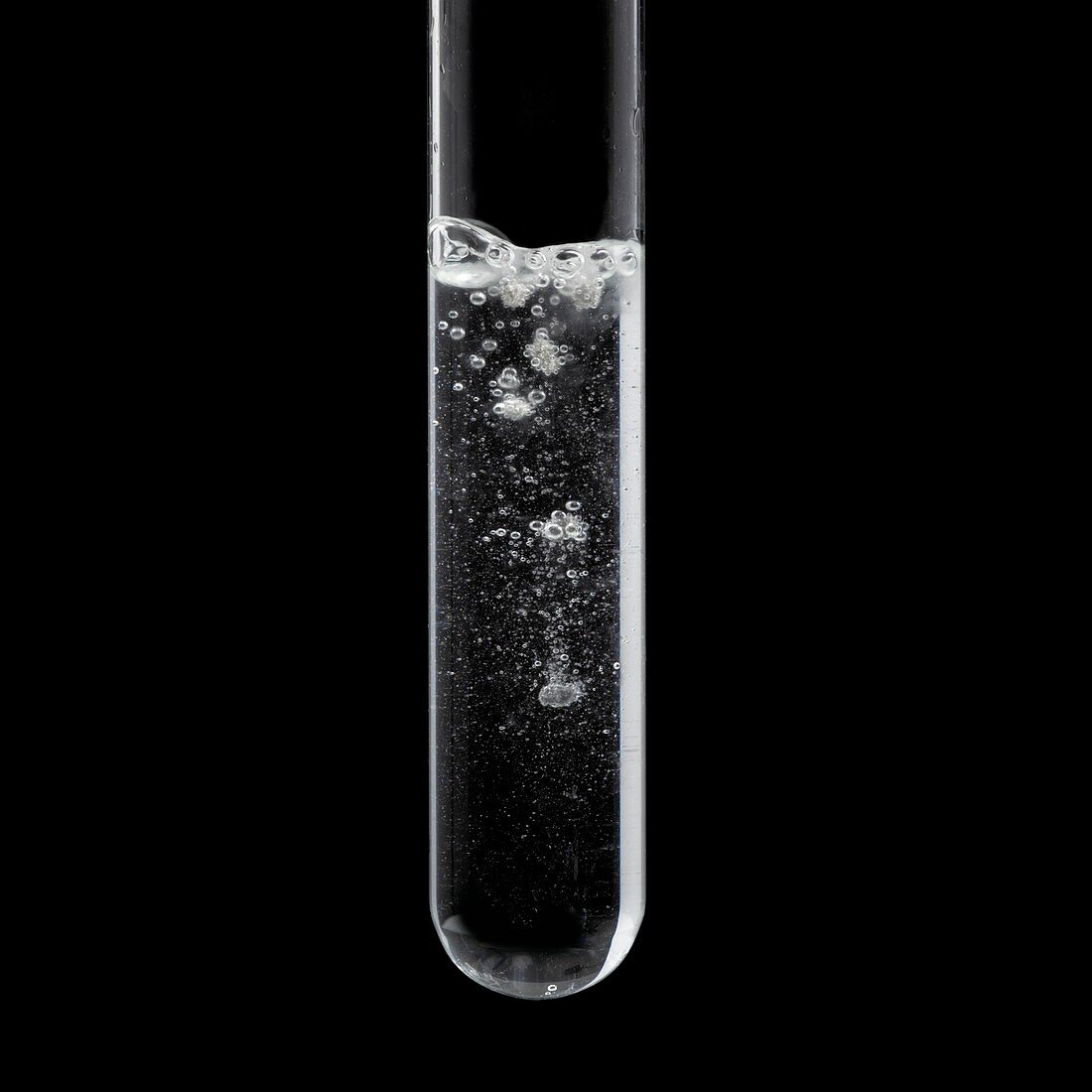
| Calcium reacting with water, initial stage. Calcium metal (Ca) reacts vigorously with water (H2O), producing hydrogen gas (H2) bubbles and slightly soluble calcium hydroxide (Ca(OH)2): Ca + H2O -> Ca(OH)2 + H2. Here, a piece of calcium has been dropped into a test tube with water. Calcium first sinks (as seen here) but then bubbles up due to the hydrogen gas produced. Calcium hydroxide is later seen as white precipitate (see C040/2668). | |
| Lizenzart: | Lizenzpflichtig |
| Credit: | Science Photo Library |
| Bildgröße: | 4370 px × 4370 px |
| Modell-Rechte: | nicht erforderlich |
| Eigentums-Rechte: | nicht erforderlich |
| Restrictions: | - |
Preise für dieses Bild ab 15 €
Universitäten & Organisationen
(Informationsmaterial Digital, Informationsmaterial Print, Lehrmaterial Digital etc.)
ab 15 €
Redaktionell
(Bücher, Bücher: Sach- und Fachliteratur, Digitale Medien (redaktionell) etc.)
ab 30 €
Werbung
(Anzeigen, Aussenwerbung, Digitale Medien, Fernsehwerbung, Karten, Werbemittel, Zeitschriften etc.)
ab 55 €
Handelsprodukte
(bedruckte Textilie, Kalender, Postkarte, Grußkarte, Verpackung etc.)
ab 75 €
Pauschalpreise
Rechtepakete für die unbeschränkte Bildnutzung in Print oder Online
ab 495 €
Keywords
- ausgeschnitten,
- Ausschnitte,
- Bildung,
- Bildungs-,
- Ca (OH) 2,
- Chemie,
- chemisch,
- Demonstration,
- demonstrierend,
- einer,
- Experiment,
- H2,
- Labor,
- Luftblasen,
- Naturwissenschaft in der Schule,
- Niemand,
- Reagenzglas,
- reagieren,
- Reaktion,
- Reihenfolge,
- schwarzer Hintergrund,
- Serie,
- Single,
- Studioaufnahme,
- Wasser
