Hydrogen Peroxide, Catalytic Decomposition, 1 of 2
Bildnummer 12639579
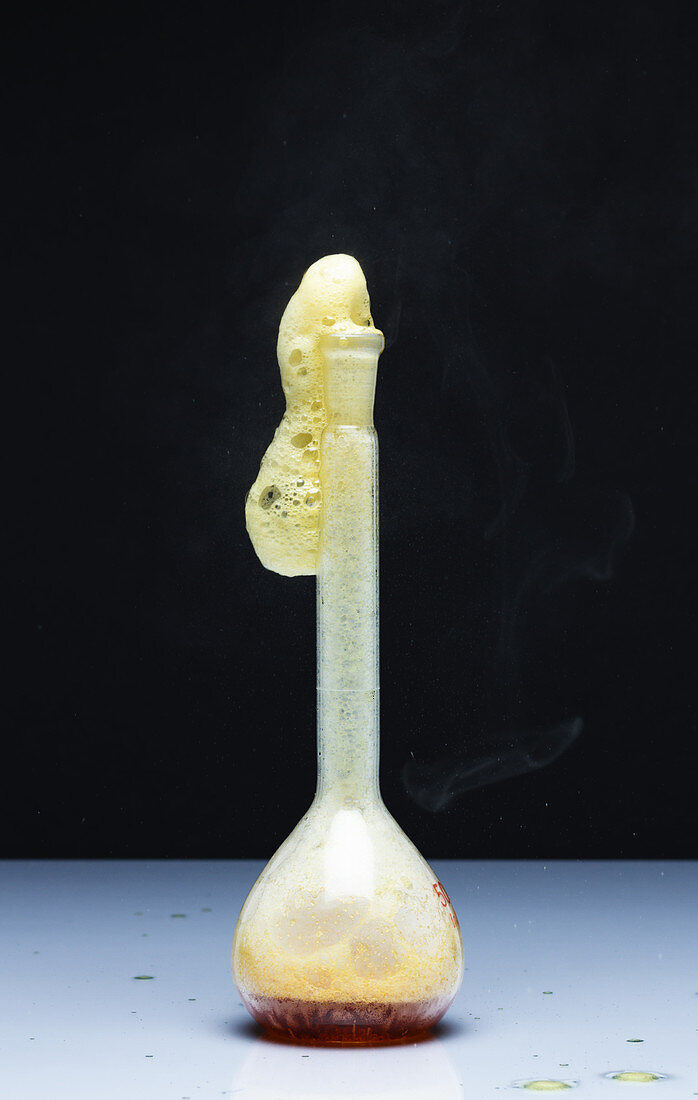
| Catalytic decomposition of hydrogen peroxide, 1 of 2. A 30% hydrogen peroxide solution (H2O2) undergoes a very slow decomposition into water (H2O) and oxygen gas (O2) under normal conditions. The rate of decomposition is greatly increased by addition of powdered solid potassium iodide (KI) which acts as a catalyst. The decomposition of hydrogen peroxide occurs in two steps: H2O2 (aq) + I- (aq) -> H2O(l) + OI-(aq) and H2O2(aq) + OI-(aq) -> H2O(l) + O2(g) + I-(aq). | |
| Lizenzart: | Lizenzpflichtig |
| Credit: | Science Photo Library / Turtle Rock Scientific |
| Bildgröße: | 3029 px × 4773 px |
| Modell-Rechte: | nicht erforderlich |
| Eigentums-Rechte: | nicht erforderlich |
| Restrictions: | - |
Preise für dieses Bild ab 15 €
Universitäten & Organisationen
(Informationsmaterial Digital, Informationsmaterial Print, Lehrmaterial Digital etc.)
ab 15 €
Redaktionell
(Bücher, Bücher: Sach- und Fachliteratur, Digitale Medien (redaktionell) etc.)
ab 30 €
Werbung
(Anzeigen, Aussenwerbung, Digitale Medien, Fernsehwerbung, Karten, Werbemittel, Zeitschriften etc.)
ab 55 €
Handelsprodukte
(bedruckte Textilie, Kalender, Postkarte, Grußkarte, Verpackung etc.)
ab 75 €
Pauschalpreise
Rechtepakete für die unbeschränkte Bildnutzung in Print oder Online
ab 495 €
