Beryllium atom, illustration
Bildnummer 12445406
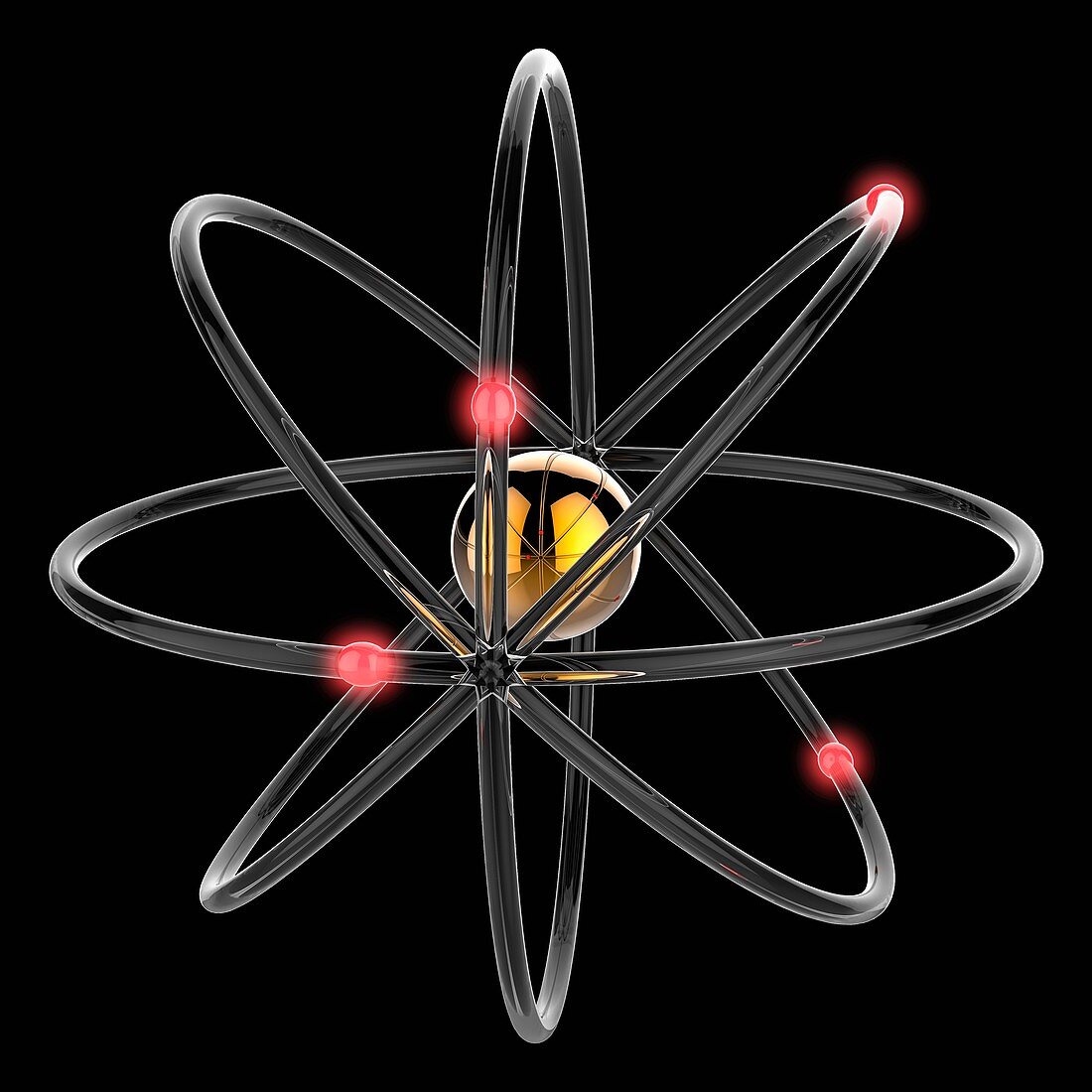
| Computer illustration of an atom of beryllium (chemical symbol Be). Its nucleus (golden sphere) contains four positively-charged protons and five neutral neutrons. Four negatively-charged electrons (red) are travelling in circular orbits around the nucleus. The charges of the protons and electrons cancel each other out so that the atom is neutral overall. Circular electron orbits were first proposed by the Danish physicist Niels Bohr in 1913. Scientists now believe that electron orbits are more eccentric and electrons inhabit a 'cloud of probability', but Bohr orbits remain a useful visualisation of an atom's structure. | |
| Lizenzart: | Lizenzpflichtig |
| Credit: | Science Photo Library / Laguna Design |
| Bildgröße: | 4180 px × 4180 px |
| Modell-Rechte: | nicht erforderlich |
| Eigentums-Rechte: | nicht erforderlich |
| Restrictions: | - |
Preise für dieses Bild ab 15 €
Universitäten & Organisationen
(Informationsmaterial Digital, Informationsmaterial Print, Lehrmaterial Digital etc.)
ab 15 €
Redaktionell
(Bücher, Bücher: Sach- und Fachliteratur, Digitale Medien (redaktionell) etc.)
ab 30 €
Werbung
(Anzeigen, Aussenwerbung, Digitale Medien, Fernsehwerbung, Karten, Werbemittel, Zeitschriften etc.)
ab 55 €
Handelsprodukte
(bedruckte Textilie, Kalender, Postkarte, Grußkarte, Verpackung etc.)
ab 75 €
Pauschalpreise
Rechtepakete für die unbeschränkte Bildnutzung in Print oder Online
ab 495 €
